The Ultimate Clinic Guide to Top Beauty Machine Manufacturer: Efficacy, Wavelengths, and ROI
Introduction: The High-Stakes Decision of Selecting a Top Beauty Machine Manufacturer
For the discerning clinic owner or medical spa director, the choice of an aesthetic device partner is not merely an operational expense—it is a strategic investment that defines treatment outcomes, patient safety, and long-term profitability. A truly top beauty machine manufacturer differentiates itself through clinical-grade engineering, validated photothermolysis mechanisms, and a demonstrable return on investment (ROI). This guide provides a data-driven framework for evaluating these systems, moving beyond marketing buzzwords to analyze the core metrics that matter: wavelength precision (755nm, 808nm, 1064nm), energy density (Fluence: 10-120 J/cm²), pulse width (ms), and epidermal protection systems compliant with Medical CE, FDA clearance, and ISO 13485 manufacturing standards.
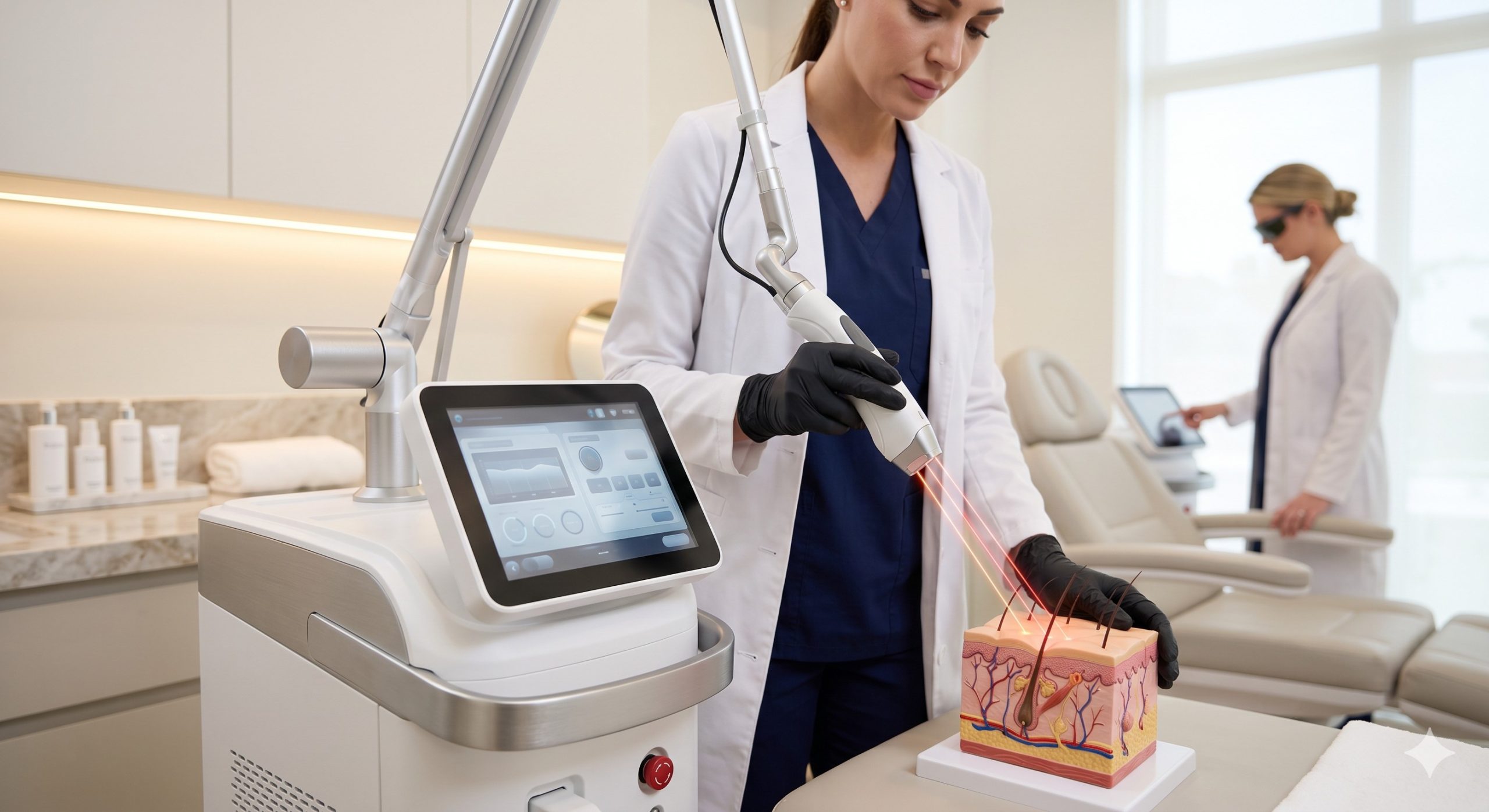
Physical Mechanisms of Action: Selective Photothermolysis and Tissue Interaction
The clinical superiority of a premium diode laser system lies in its strict adherence to the principle of selective photothermolysis. For permanent hair reduction, the target chromophore is melanin within the hair follicle. A top-tier manufacturer optimizes this by offering a tri-wavelength or dual-wavelength platform (e.g., 755nm, 808nm, 1064nm). The 755nm wavelength offers high melanin absorption for lighter skin types (Fitzpatrick I-III), while the 1064nm wavelength provides deeper penetration with reduced epidermal absorption, making it the safest option for darker skin types (Fitzpatrick IV-VI). The 808nm diode represents the ‘goldilocks’ wavelength, balancing absorption and depth. Key parameters affecting clinical clearance rates include:
- Fluence (Energy Density): Ranges from 10 to 120 J/cm². Higher fluence is required for coarse or deep follicles, but must be balanced with cooling.
- Pulse Width (Duration): Typically 5-400 ms. Shorter pulses (5-30 ms) target rapid thermal relaxation in fine hair; longer pulses (100-400 ms) are safer for large areas or darker skin.
- Spot Size: A larger spot size (e.g., 12×12 mm² to 15×15 mm²) allows for deeper photon penetration and faster treatment speeds, reducing overall session time by up to 60%.
Advanced Cooling Engineering: The Safeguard of Epidermal Protection
High fluence treatments without robust cooling risk thermal damage to the epidermis. A leading top beauty machine manufacturer integrates advanced Sapphire contact cooling with thermoelectric (TEC) modules. This system maintains the skin surface at 0°C to 4°C pre-, during, and post-laser pulse. Clinical studies confirm that sapphire cooling not only reduces pain (patient comfort scores >8/10) but also allows the clinician to safely use therapeutic fluences up to 40% higher than with traditional cooling gels or air cooling alone. Look for systems with real-time skin temperature monitoring and automatic shut-off safety interlocks.
Critical Technical Specifications Matrix
The following table outlines the non-negotiable technical metrics that define a medically credible device. Always verify these specifications against the manufacturer’s ISO 13485 certified documentation.
| Key Parameter | Technical Specification |
|---|---|
| Laser Type / Wavelengths | Medical Diode Laser: 755nm (Alexandrite), 808nm (Diode), 1064nm (Nd:YAG) – Selectable or Simultaneous |
| Energy Density (Fluence) | 10 – 120 J/cm² (Adjustable in 1 J/cm² increments) |
| Pulse Width (Duration) | 5 – 400 ms (Multi-pulse capability for safety) |
| Spot Size Options | 12×12 mm², 15×15 mm², and 10×30 mm² (fast scanning) |
| Cooling System | Active Sapphire Contact Cooling with TEC module (0°C to 4°C surface temperature) |
| Repetition Rate | Up to 10 Hz (10 pulses per second) for rapid coverage |
| Regulatory Compliance | Medical CE (Class IIb), FDA 510(k) Cleared, ISO 13485:2016 Manufactured |
| Handpiece Durability | 1 million to 5 million shot lifespan (verified test cycles) |
Treatment Parameters and Fitzpatrick Skin Typing Protocol
Clinical excellence requires matching device parameters to individual patient phenotypes. A standardized protocol for a high-output 808nm system includes:
Fitzpatrick Skin Types I-III
- Wavelength: 755nm or 808nm
- Fluence: 10-14 J/cm² (fine hair) to 16-20 J/cm² (coarse hair)
- Pulse Width: 5-30 ms
- Spot Size: 12×12 mm² or larger
Fitzpatrick Skin Types IV-VI
- Wavelength: 1064nm (primary) or 808nm with conservative settings
- Fluence: 8-12 J/cm² (test spot essential, starting low)
- Pulse Width: 100-400 ms
- Cooling: Maximum sapphire cooling (0-2°C) with extended pre-cooling.
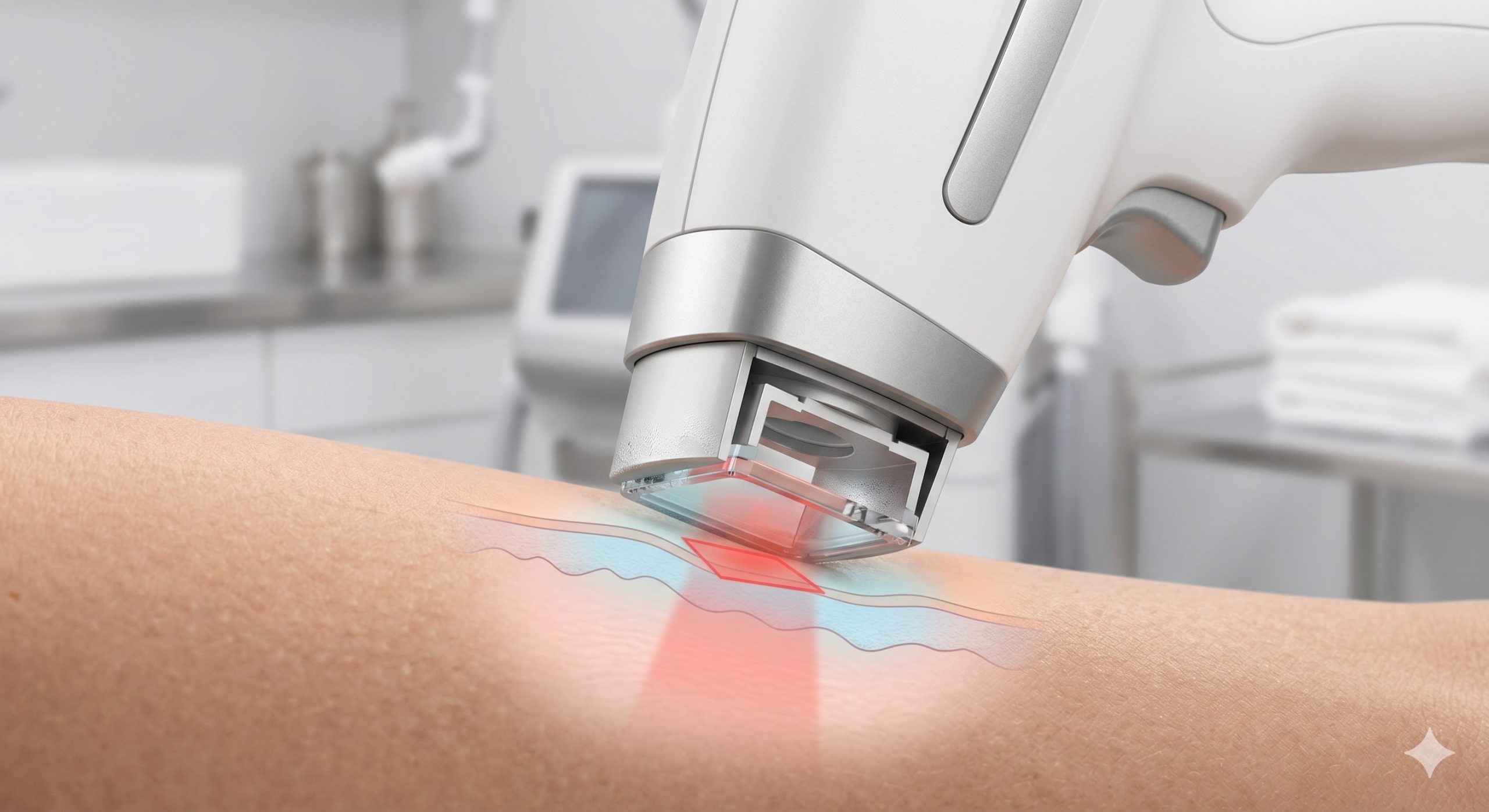
Clinic ROI and Profitability Analysis
Beyond clinical metrics, the economic case for a top-tier device is compelling. Consider a high-volume clinic performing 20 hair removal sessions per week. At an average price of $250 per session (full legs or back), weekly revenue is $5,000. Annually (50 weeks), gross revenue is $250,000. The cost of ownership includes:
- CapEx (Device Purchase): $40,000 – $80,000 (premium diode laser). Depreciated over 5 years: $8,000 – $16,000/year.
- OpEx (Consumables & Maintenance): Low for diode lasers; most quality systems offer 1-5 million shots per handpiece before replacement. Energy and cooling maintenance: ~$500/year.
- Net Profit Margin: 70-85% after operator costs and consumables.
ROI break-even for a premium top beauty machine manufacturer device is typically achieved within 4 to 9 months of clinical use, assuming moderate patient volume. Furthermore, offering laser services dramatically increases average patient lifetime value (LTV), as hair removal typically requires 6-8 sessions per patient.
Compliance and Safety Standards Verification
Before procurement, mandate that the manufacturer provide verifiable proof of Medical CE (Class IIb or III), FDA 510(k) clearance for specific indications (e.g., permanent hair reduction), and factory ISO 13485:2016 certification. These registrations are not marketing fluff; they are legal attestations to the device’s safety, efficacy, and manufacturing quality. Clinics using non-certified devices face significant liability risks, insurance invalidation, and potential closure. Always cross-reference the device name on the official FDA and EUDAMED databases.
Conclusion: Strategic Procurement for Clinical Excellence
Selecting a top beauty machine manufacturer is a multidimensional decision. Prioritize vendors who provide transparent technical specifications (wavelengths, fluence, pulse width, spot size), medically certified cooling systems, and full regulatory documentation (Medical CE, FDA, ISO 13485). The initial higher CapEx for a clinically superior diode laser system (755/808/1064nm) is offset by rapid ROI, high patient satisfaction, and low lifetime cost of ownership. For the modern med spa, a laser platform from a verified top manufacturer is not an expense—it is the engine of sustainable profitability.