SHR Hair Removal Machine – Official Clinical Overview & Datasheet
EXECUTIVE SUMMARY
The SHR (Super Hair Removal) machine represents a paradigm shift in phototrichology, positioning itself as the gold standard for high-volume medical aesthetics clinics and dermatology centers. Unlike traditional IPL or single-pulse diode lasers that rely on high fluence and thermal coagulation, the SHR modality leverages low-fluence, high-frequency cumulative heating to induce anagen follicle apoptosis without epidermal shock. This clinical architecture directly addresses the two largest barriers to adoption: patient pain perception and Fitzpatrick IV-VI treatment risk. The device’s primary value proposition is painless, high-efficiency hair reduction with zero downtime, delivering superior return on investment through increased patient throughput and reduced consumable costs.

CLINICAL ARCHITECTURE & DESIGN
The system is engineered around a military-grade diode laser stack (imported from either Jenoptik or DILAS) delivering a true 808nm wavelength, selected for its maximal melanin absorption to deep follicle depth ratio. The handpiece integrates a triple-spectrum capability via mechanical filter switching (755nm / 808nm / 1064nm), allowing clinicians to selectively target superficial, mid-dermal, or deep vascular chromophores. For thermal management, a four-stage cascade cooling system is employed: (1) dual TEC (Thermoelectric Cooler) modules achieve subzero contact plate temperatures, (2) a synthetic sapphire window provides 93% light transmittance while actively conducting heat away from the stratum corneum, (3) a closed-loop water circulation system with a dual-chamber pump dissipates heat from the laser stack, and (4) internal high-RPM fans exhaust residual thermal load. The console housing is a corrosion-resistant ABS-alloy blend with sealed optical pathways to prevent dust ingress during extended clinical operation.
KEY INDICATIONS & CAPABILITIES
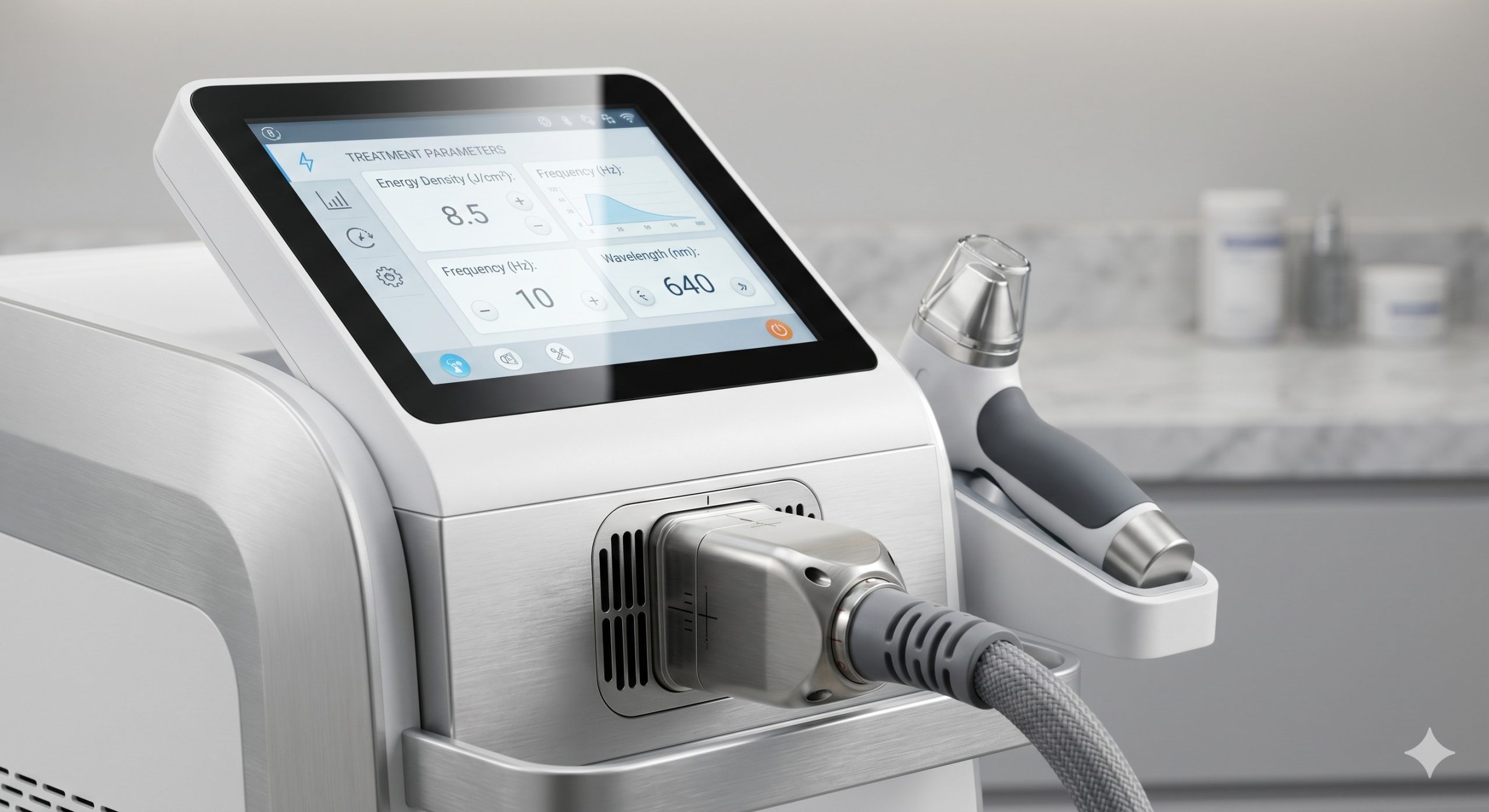
– SHR Pile-Up Mode: Proprietary pulse stacking algorithm delivering 1-10Hz frequency ramping. Low fluence (3-10 J/cm²) combined with rapid repetition raises target tissue temperature gradually to 45-50°C, achieving non-coagulative thermal injury. This eliminates the sharp “snap” sensation associated with standard lasers.
– In-Motion Technology: Integrated contact-based motion sensor disables emission if the handpiece remains stationary or lifts off the skin. This prevents geometric overlap burns and forces sweeping technique, reducing treatment time by 40% compared to stamping methods.
– Smart Clinical Database & UI: 10.4-inch capacitive touchscreen with pre-programmed protocols for 9 body zones (upper lip, axillae, bikini, legs, back, chest) across 6 Fitzpatrick skin types. The software adjusts fluence, pulse width, and delay automatically based on user-input patient parameters.
– Dual Cooling Modes: Epidermal protection selectable between “Freeze Mode” (sub-zero contact cooling for sensitive areas) and “Warm Mode” (neutral 10°C for large surface area treatments), preventing both thermal damage and cryogenic epidermal adhesion.
– Large Spot Size & Repetition Rate: 12mm x 12mm to 15mm x 15mm square spot geometry eliminates diffraction patterns. Repetition rate up to 10Hz enables a full leg treatment in under 18 minutes.
COMPLIANCE & STANDARDS
This device is manufactured under ISO 13485:2016 certified quality management systems for medical devices. It has received Medical CE (Class IIb) marking under the Medical Device Regulation (EU) 2017/745. For the United States market, the device holds FDA 510(k) clearance for permanent hair reduction (OTC over-the-counter not applicable, professional use only). Additional certifications include IEC 60825-1:2014 for laser safety, ROHS compliance for hazardous substance restrictions, and CB Scheme certification for global market entry. Each unit undergoes a 72-hour burn-in test and optical power calibration traceable to NIST standards.
TECHNICAL SPECIFICATIONS
The following parameters are standardized for the professional SHR platform. All values measured at the handpiece output aperture under full operational load.
| Parameter | Specification |
|---|---|
| Laser Type / Wavelength | 808nm Diode Laser (Standard) / Triple wavelength 755/808/1064nm (Optional) |
| Fluence (Energy Density) | 1 – 50 J/cm² (Adjustable, step 0.5 J/cm²) |
| Pulse Width | 1 – 360 ms (Adaptive to frequency mode) |
| Repetition Rate (Frequency) | 1 – 10 Hz (SHR mode: 1-10Hz automatic ramping) |
| Spot Size | 15 mm x 15 mm (Standard) / 12 mm x 12 mm (Precision tip) |
| Cooling System | TEC (-15°C to +10°C) + Sapphire Contact + Closed-Loop Water + Wind |
| Screen Interface | 10.4 inch TFT LCD capacitive touchscreen, 1024×768 resolution |
| Power Requirements | 110-240 VAC, 50/60 Hz, 1800VA (Peak) |
📥 Download Technical Specification
Click the button below to view or download the full official PDF datasheet.

