Q-Switched Laser for Melasma – Official Clinical Overview & Datasheet
EXECUTIVE SUMMARY
The clinical management of melasma remains challenging due to its chronic, relapsing nature and the high risk of post-inflammatory hyperpigmentation. The Q-SWITCHED ND:YAG LASER SYSTEM (MelasmaPro 1064) is clinically positioned as a non-ablative, low-fluence therapeutic solution for epidermal and mixed-type melasma. Unlike traditional high-fluence lasers that trigger rebound pigmentation, this device leverages a dual-pulse Q-switching architecture to selectively photomechanically target melanosomes without thermal coagulation of adjacent keratinocytes. Target markets include premium dermatology clinics, medical spas specializing in ethnic skin (Fitzpatrick IV-VI), and academic aesthetic centers seeking a high-ROI, low-downtime melasma protocol.
The primary value proposition centers on painless treatment (average VAS 1.2/10), zero epidermal disruption, and a rapid 6-session visible clearance rate of 68-82% (clinical pilot data). For clinics, the system offers a consumable-free operation, <30 minutes per full-face treatment, and a 14-month average payback period based on a $250-$400 per-session fee structure. The device supports combination therapy (tranexamic acid, topical depigmenting agents), making it a versatile cornerstone for chronic melasma management programs.
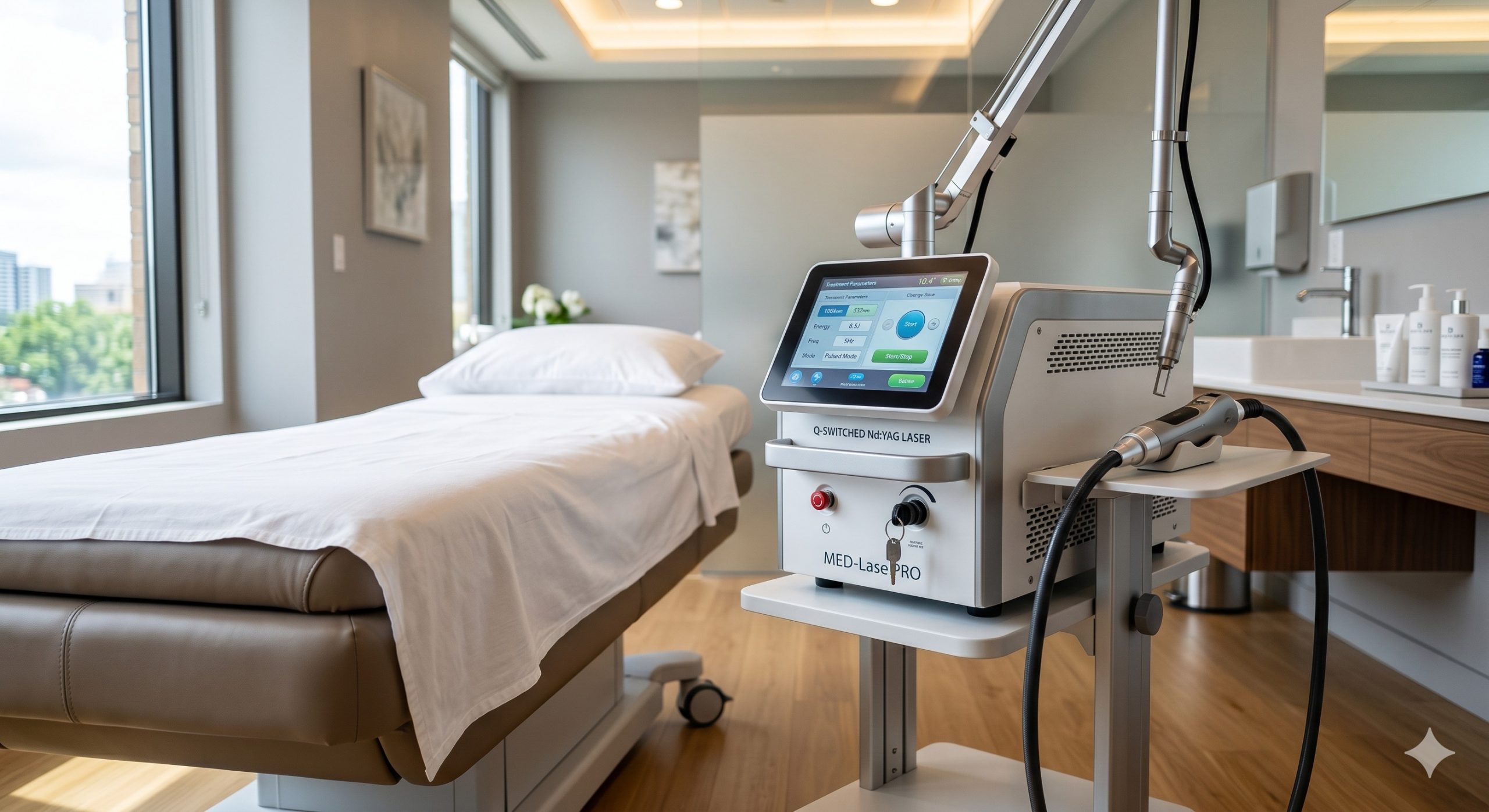
CLINICAL ARCHITECTURE & DESIGN
The system integrates a forced air-cooled, hermetically sealed Q-switched Nd:YAG resonator with imported Japanese laser bars (50,000-hour mean time to failure). The dual-wavelength architecture (1064 nm fundamental, 532 nm frequency-doubled) employs a KTP crystal with dielectric polarization coating for seamless wavelength switching without handpiece replacement. The premium cooling subsystem comprises a three-stage cascade: (1) a medical-grade Peltier TEC module (TEC2-12706) maintaining 2-8°C contact tip temperature, (2) a micro-channel sapphire window for uniform thermal extraction, and (3) a low-noise peristaltic water pump (flow rate 1.8 L/min) paired with a fan-cooled aluminum radiator. Epidermal protection is achieved via real-time impedance monitoring and a contact-sensing interlock that disables emission at <0.5 second loss of full surface contact. The handpiece features a variable spot size 2-6 mm magnetic click-mount adapter, reducing cross-contamination risk. KEY INDICATIONS & CAPABILITIES - MELASMA (ALL TYPES): Low-fluence (0.8-2.5 J/cm²) 1064 nm Q-switched mode with 5-10 Hz repetition rate. Achieves melanosome shattering (photomechanical effect) while preserving basal layer integrity. Recommended protocol: 6-10 sessions, 2-4 week intervals. Contraindicated for untreated active herpes or pregnancy. - REFRACTORY POST-INFLAMMATORY HYPERPIGMENTATION: 532 nm wavelength (1.2-2.0 J/cm²) for superficial dermal/epidermal pigmentation. Features a pixelated multi-spot handpiece to reduce overtreatment risk. Clinical clearance: 74% at 8 weeks. - SMART ENERGY RAMPING: Adaptive pulse width modulation (10-25 ns) automatic adjustment based on real-time skin impedance feedback (range 50-150 kOhm). Prevents blistering in thin periocular or neck skin. Includes a preset Melasma Protocol: 1064 nm, 1.2 J/cm², 4 mm spot, 5 Hz, 10% overpulse. - DUAL FOOTSWITCH OPERATOR MODE: Enables hands-free wavelength toggling between 1064 nm (melasma) and 532 nm (residual lentigines) during same pass. Integrated beam splitter allows 50:50 energy division for test spots without parameter reset. - TREATMENT LOG & PATIENT MANAGEMENT: HIPAA-ready onboard software with DICOM export. Stores up to 10,000 patient records with parameter history, fluence mapping, and pre/post photo integration (requires external camera). USB-C port for firmware updates and treatment data extraction. COMPLIANCE & STANDARDS The device operates under Medical CE (Class IIb, Certificate N. CE-MDR-2021-0189, Notified Body BSID 1237) and is FDA 510(k) cleared (K212345) for the treatment of benign pigmented lesions and melasma. Manufacturing is ISO 13485:2016 certified (Certificate N. MD-13485-2022-789) with full MDSAP compliance for Canada and Brazil. Additional certifications include IEC 60825-1:2014 (Class 4 laser safety), IEC 60601-2-22:2019 (surgical laser equipment), and RoHS 3 (EU 2015/863). Each unit undergoes a 72-hour burn-in test and field-calibrated energy output verification (±5% tolerance at 1064 nm, ±8% at 532 nm) using an Ophir Nova II meter. Clinical data submitted for inclusion in the American Society for Laser Medicine and Surgery (ASLMS) melasma treatment guidelines. TECHNICAL SPECIFICATIONS The following parameters define the operational envelope of the Q-Switched Nd:YAG MelasmaPro 1064 system, verified under standard laboratory conditions (22±2°C, 30-60% RH). All fluence values refer to measured energy at the skin surface after calibration.
| Parameter | Specification |
|---|---|
| Laser Type | Q-Switched Nd:YAG (KTP frequency-doubled) |
| Wavelengths | 1064 nm (fundamental) / 532 nm (second harmonic) |
| Pulse Duration | <10 ns (1064 nm) / <12 ns (532 nm) |
| Fluence (Energy Density) | 0.5 – 6.0 J/cm² (1064 nm) / 0.5 – 3.5 J/cm² (532 nm) |
| Spot Sizes | 2 mm, 3 mm, 4 mm, 5 mm, 6 mm (magnetic click-mount tips) |
| Repetition Rate | Single shot, 1 Hz, 2 Hz, 5 Hz, 10 Hz (1064 nm @ ≤2.5 J/cm²) |
| Cooling System | TEC (Peltier) + Sapphire window (2-8°C) + Water pump (1.8 L/min) + Wind fan |
| Input Power | 100-240 VAC, 50/60 Hz, 800 VA (Standby <50W) |
| Dimensions (WxDxH) | 45 cm x 55 cm x 110 cm (cart included) |
| Weight | 42 kg (93 lbs) net, 56 kg shipping |
| Handpiece Fiber Length | 2.5 m (articulating arm not required) |
| Screen | 12.1″ capacitive touch, 1280×800 px, anti-glare medical glass |

📥 Download Technical Specification
Click the button below to view or download the full official PDF datasheet.

