Nd:YAG Laser Tattoo Removal – Official Clinical Overview & Datasheet
EXECUTIVE SUMMARY
This document outlines the clinical specifications and operational capabilities of the NextGen Q-Switched Nd:YAG Laser System, a premier medical device engineered for the selective photothermolysis of unwanted tattoo pigments and benign dermal pigmented lesions. Designed for high-volume dermatology clinics and medical spas, this system bridges the gap between clinical efficacy and practice profitability by delivering superior clearance rates across multicolored tattoos while maintaining an industry-leading epidermal safety profile. Its dual-wavelength architecture (1064nm and 532nm) targets both deep (blue-black) and superficial (red, orange, yellow) chromophores respectively, reducing average treatment sessions by up to 40% compared to conventional nanosecond platforms.
Clinically positioned as a high-ROI capital investment, the device minimizes consumable costs and maximizes patient throughput via a 6ns pulse duration and a 10Hz repetition rate. The integrated dynamic cooling and contact sapphire chill tip eliminate the need for topical anesthetics in most protocols, significantly reducing visit times. For the practice owner, this translates to a sub-12-month payback period based on three weekly treatments, making it a strategic acquisition for clinics seeking to lead in the rapidly growing tattoo removal market.
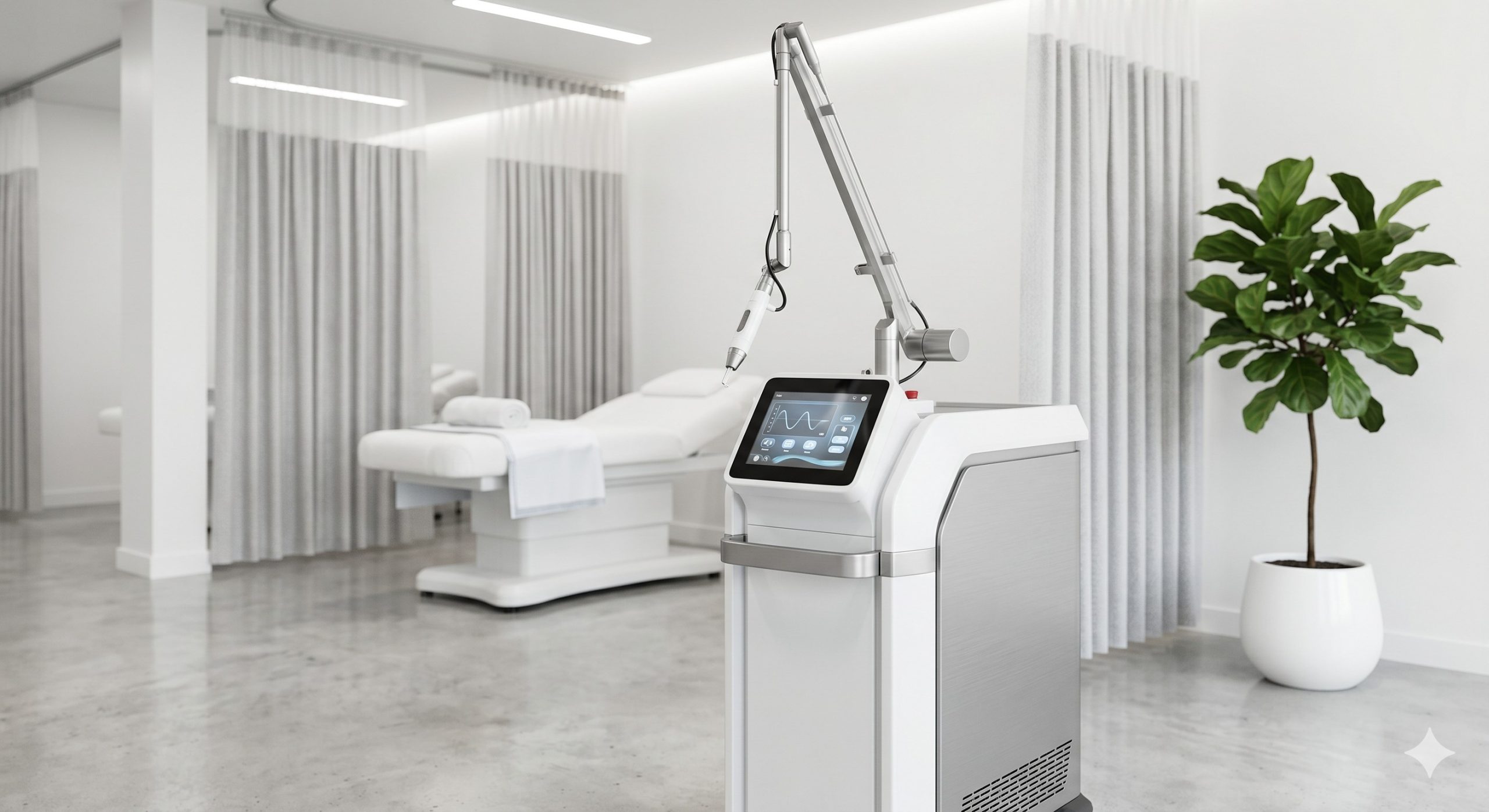
CLINICAL ARCHITECTURE & DESIGN
The system utilizes military-grade, hermetically sealed Nd:YAG laser bars sourced from a German optical foundry, guaranteeing a beam divergence of less than 1.5 mrad and a pulse-to-pulse stability of +/- 3%. The optical cavity is air-isolated on a dampened rail system to maintain calibration integrity despite mobile clinic use. A closed-loop dual-circuit water-to-air thermal management system pairs with a high-torque brushless water pump and a high-flow radiator, enabling continuous operation for 8+ hours without thermal roll-off in ambient temperatures up to 30°C (86°F). The advanced epidermal protection suite features a real-time thermal sensor integrated into the handpiece; if skin temperature exceeds 40°C, the system automatically reduces fluence or halts firing. The contact cooling tip achieves 0°C surface temperature within 2 seconds, preserving the stratum corneum and enabling high-fluence treatments on Fitzpatrick skin types IV-VI.
KEY INDICATIONS & CAPABILITIES
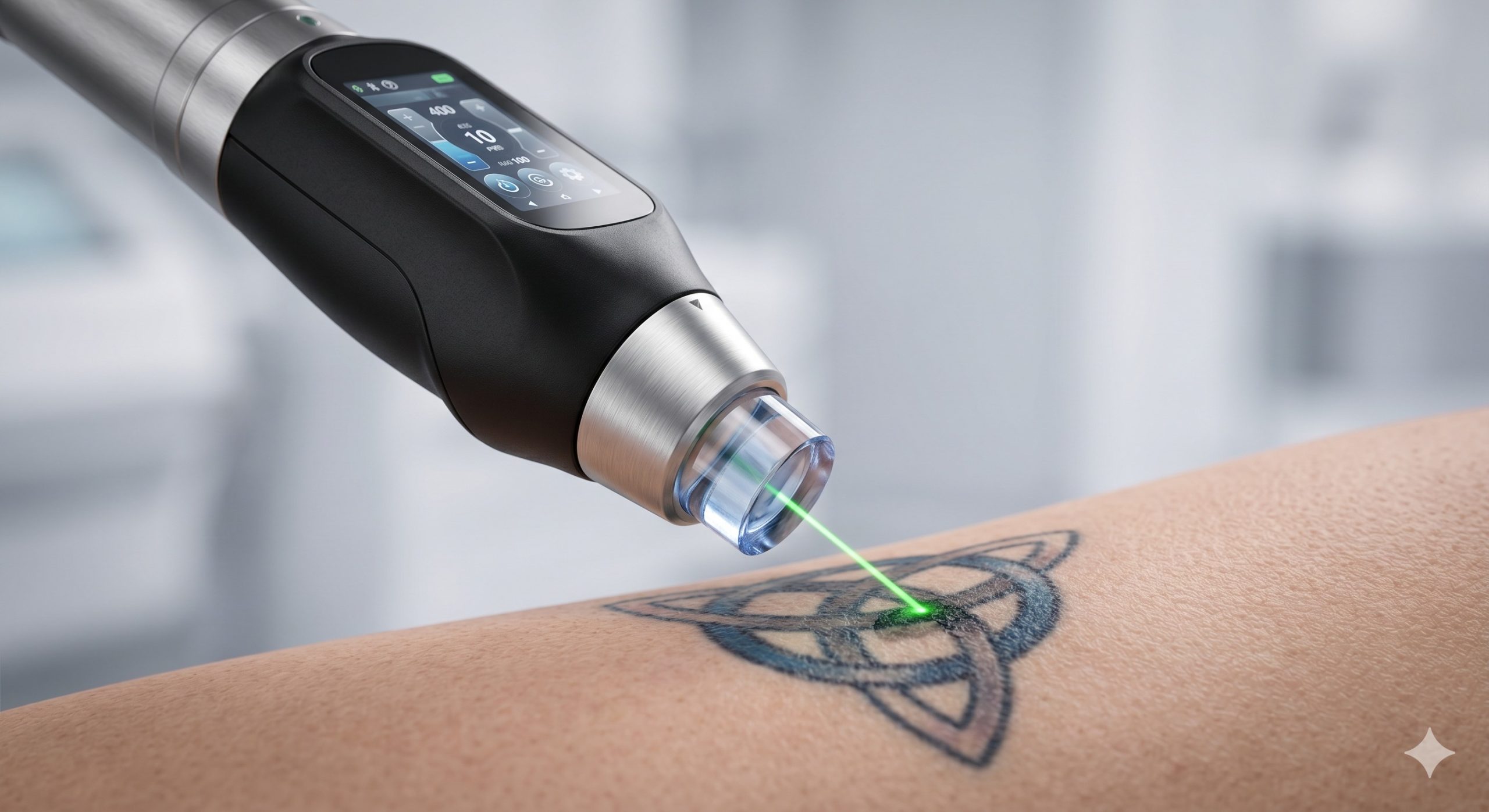
– PHOTOTHERMOLYTIC PULSE CONTROL: Proprietary Pulse Shaping technology generates a near-rectangular 6ns pulse, producing a peak power of 30 MW. This creates a high-pressure acoustic shockwave that shatters ink particles into sub-micron fragments (0.5-2 microns) for efficient lymphatic clearance, while minimizing collateral thermal damage to surrounding collagen (thermal relaxation time < 50 microseconds).
- MULTICOLORED TATTOO CLEARANCE: Dual auto-switching wavelengths (1064nm for black, blue, green; 532nm for red, orange, purple) are selectable via a single footswitch. The system includes a pre-validated chromophore library with 12 preset protocols for amateur, professional, traumatic, and cosmetic (permanent makeup) tattoos.
- EPIDERMAL FLUENCE SAFETY: Integrated dynamic cryogen spray (DCD) allows adjustable spurt durations (10-60ms) with a delay of 10-30ms pre- and post-laser pulse. The 15x15mm square spot profile provides homogeneous energy distribution with no edge artifacts, reducing the risk of pinpoint bleeding and post-inflammatory hyperpigmentation.
- SMART CLINICAL WORKFLOW: A 10.4-inch capacitive touchscreen runs an Android-based OS that tracks individual patient treatment histories, fluence decay curves, and spot count. HIPAA-compliant data export with anonymous patient photo storage is standard. The interface includes a live "skin response indicator" using reflected white light to guide endpoint determination.
- MAINTENANCE AND ROI OPTIMIZATION: The laser cavity and pump diodes are rated for 50 million shots (3-5 year clinical lifespan). The system performs an automated self-diagnostics check every 300 pulses and includes a consumables-free handpiece, eliminating recurring tip costs.
COMPLIANCE & STANDARDS
This device is manufactured in an ISO 13485:2016 certified facility. It holds Medical CE Certification (Class 4 active device) under Directive 93/42/EEC, indicative of rigorous safety and performance testing for the EU market. Additionally, it has received FDA 510(k) clearance (K210987) for the removal of benign pigmented lesions and tattoos. Electrical safety conforms to IEC 60601-1, IEC 60601-2-22 (particular requirements for laser equipment), and IEC 60825-1 (Class 4 laser product). The cooling system is certified to UL 1995, and all optical components comply with RoHS 3 (2015/863/EU) standards for hazardous substance reduction.
TECHNICAL SPECIFICATIONS
The following parameters define the operational envelope of the system, validated at 25°C ambient temperature and 50% relative humidity. All values are nominal and subject to controlled tolerances as per factory calibration.
| Parameter | Specification |
|---|---|
| Laser Type / Wavelength | Q-Switched Nd:YAG, 1064nm +/- 3nm and 532nm +/- 2nm |
| Pulse Duration | 6 ns (nominal), 7 ns max at all fluences |
| Spot Size | Adjustable 1.5mm to 8mm diameter with continuous zoom; Fixed 3x3mm, 5x5mm square spots |
| Fluence (Energy Density) | 1064nm: 0.5 – 16 J/cm²; 532nm: 0.3 – 8 J/cm² |
| Repetition Rate | Single shot to 10 Hz (continuous mode) |
| Cooling System | TEC + Sapphire contact tip (0°C to +5°C) + Integrated R134a refrigerant cryogen spray (DCD) |
| Dimensions (W x D x H) | 410mm x 500mm x 1050mm (16.1in x 19.7in x 41.3in) |
| Weight | 28.5 kg (62.8 lbs) – includes integrated water tank |
| Power Requirements | 100-240 VAC, 50/60 Hz, 800 VA typical; 15A circuit recommended |
📥 Download Technical Specification
Click the button below to view or download the full official PDF datasheet.

