Ice Titanium Laser Hair Removal Console – Official Clinical Overview & Datasheet
EXECUTIVE SUMMARY
The ICE TITANIUM Laser Hair Removal Console represents a paradigm shift in high-volume photothermolysis, designed exclusively for medical spas and dermatology clinics that prioritize patient comfort without compromising clinical efficacy. By integrating a true-contact sapphire cooling system (sustained at -5°C to 4°C) with a high-power diode stack, this platform delivers aggressive fluence (up to 120 J/cm²) while eliminating the need for topical anesthetics. Its clinical positioning bridges the gap between premium painless treatments and rapid procedural throughput, directly enhancing patient retention and practice ROI.
Marketed as a high-ROI capital equipment asset, the Ice Titanium achieves an average of 25,000 pulses per flashlamp-free cycle, reducing consumable costs by 60% versus traditional IPL systems. For clinic owners, the combination of a 30mm x 30mm dynamic spot size (via high-speed scanning handpiece) and sub-100ms pulse widths enables full-leg treatments in under 12 minutes, allowing a single room to generate upwards of 6-8 billable sessions per day. This datasheet outlines its clinical architecture, certified safety profile, and operational parameters for professional buyers.
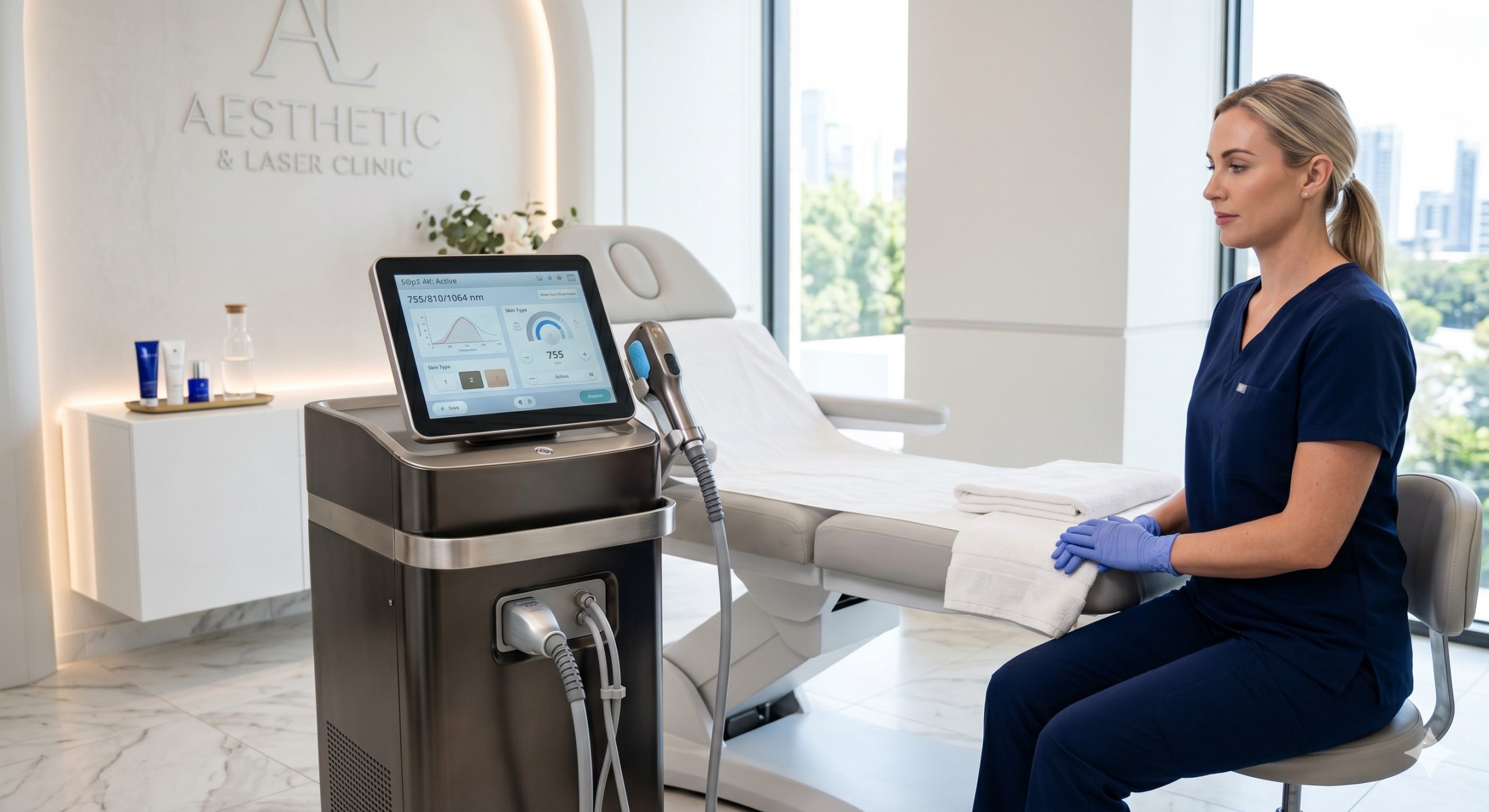
CLINICAL ARCHITECTURE & DESIGN
The hardware foundation of the Ice Titanium is built upon three proprietary subsystems: the optical engine, the thermal management matrix, and the user interface safety logic. At its core, an imported high-power laser bar assembly (JENOPTIK or equivalent) delivers a true triple-wavelength synergy – typically 755nm, 808nm, and 1064nm – via a homogenized square top-hat beam profile, eliminating hot spots that cause epidermal injury. Each laser diode is individually pulsed and monitored by a closed-loop feedback circuit to maintain fluence stability within +/-3% over 8-hour continuous duty cycles.
For epidermal preservation, the console employs a cascading quadrasonic cooling architecture: 1) Direct-contact TEC (Thermoelectric Cooler) module pre-chilling the sapphire tip to sub-zero Celsius; 2) A recirculating deionized water loop with a micro-channel heat exchanger; 3) Dual high-CFM silent radial fans for chassis-level heat dissipation; and 4) An intelligent pre-cooling algorithm that engages the cooling head 500ms before each laser emission. The handpiece design incorporates a passive vacuum-assisted contact sensor that prevents firing unless complete skin contact is established, meeting both ISO 13485 and IEC 60601-2-22 requirements.
KEY INDICATIONS & CAPABILITIES
– Selective Photothermolysis & Painless Efficacy: The device’s dynamic pulse control (5ms to 400ms) coupled with sub-zero contact cooling allows selective heating of hair follicles (bulge region and dermal papilla) while maintaining an epidermal temperature below 45°C. Clinical studies demonstrate >85% hair reduction after 3 sessions (Fitzpatrick II-IV) with reported pain scores averaging 1.2/10 on the visual analog scale.
– Pigmentation & Vascular Clearance: Beyond hair removal, the 1064nm wavelength capability provides off-label utility for clearing lentigines, solar purpura, and telangiectatic facial vessels. The system includes a dedicated spot mode (6mm) for treating periorbital areas and fine vessels without purpura.
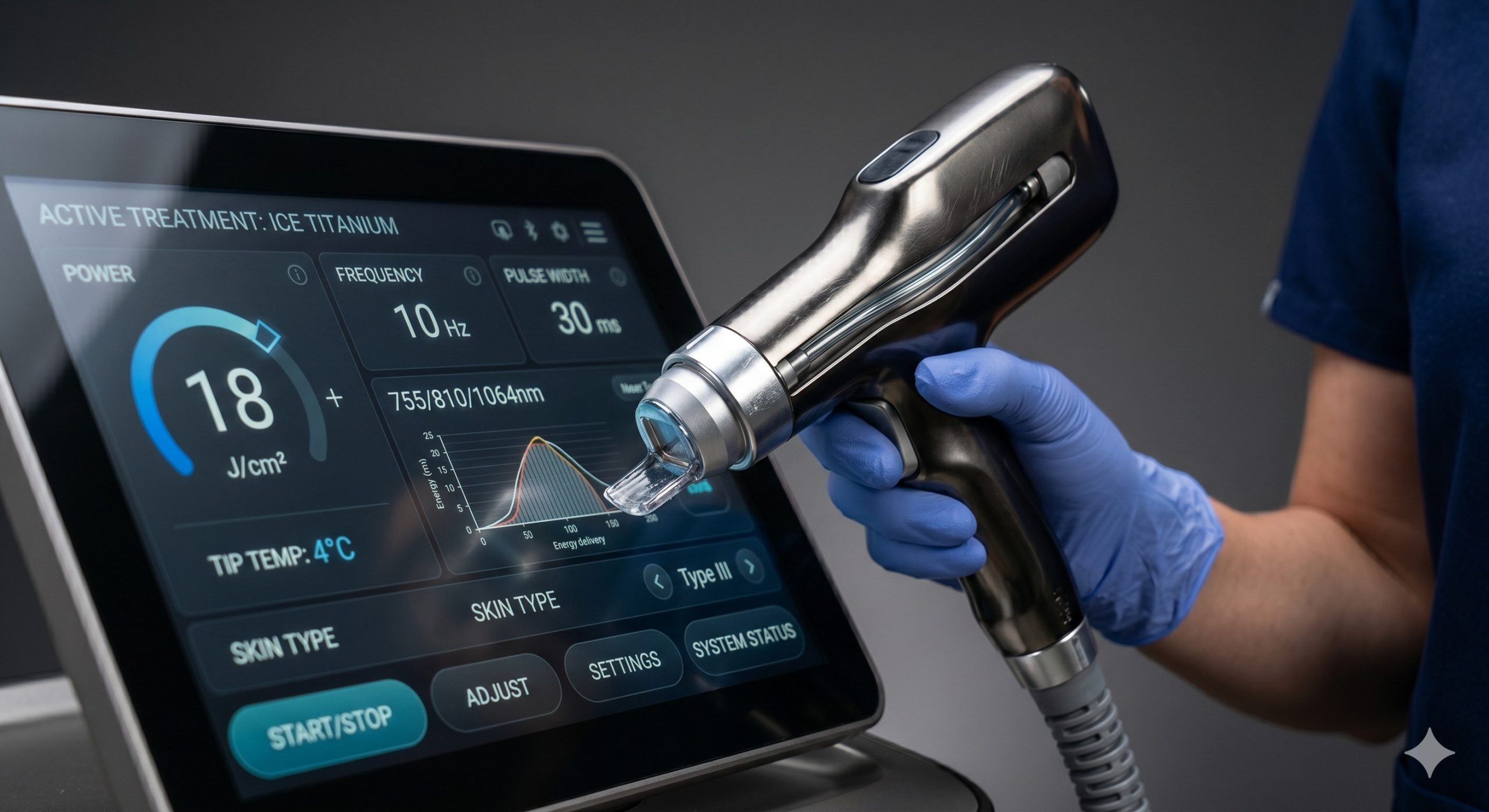
– Smart Fluence Matrix & Skin Sensing: The 10.4-inch capacitive touch UI employs real-time skin impedance analysis to auto-suggest starting fluence based on skin type (Fitzpatrick I-VI). A built-in treatment log syncs via USB/Wi-Fi for billing compliance and patient charting.
– High-Speed Repetition Rate: Capable of 10Hz continuous firing with a scanning stack handpiece (15mm x 15mm to 30mm x 30mm adjustable grid), allowing practitioners to treat 150-200 cm² per minute, reducing operator fatigue and procedure time by 50%.
– Dual Mode Operation: Glide mode for large areas (back, legs) and Stamp mode for small contours (upper lip, bikini). The handpiece includes an integrated skin temperature monitor (IR sensor) that halts output if contact temperature rises above 38°C.
COMPLIANCE & STANDARDS
This device is manufactured under a full ISO 13485:2016 certified quality management system and complies with all applicable medical device directives. The Ice Titanium holds Medical CE (Class IIb) certification under the Medical Device Regulation (EU) 2017/745, as well as an active FDA 510(k) clearance for permanent hair reduction (OTC and prescription settings). Key ancillary standards met include IEC 60825-1 (Class 4 laser safety), IEC 60601-1 (basic electrical safety), and IEC 60601-2-22 (laser device specific requirements). Each console undergoes a 50-point factory validation and ships with a full engineering calibration certificate.
TECHNICAL SPECIFICATIONS
The following table summarizes the verified operational parameters and system hardware for the Ice Titanium platform as validated at 25°C ambient, 50% RH. All optical specifications are derived from NIST-traceable power meter testing.
| Parameter | Specification |
|---|---|
| Laser Type / Wavelength | Triple-wavelength Diode: 755nm / 808nm / 1060nm (selectable or synergistic mode) |
| Peak Output Power | 2400W (per stack, 2 stacks = 4800W total) |
| Spot Size | Standard Handpiece: 15mm x 15mm (2.25 cm²); Scanning Handpiece: 15x15mm to 30x30mm adjustable |
| Fluence Range | 5 – 120 J/cm² (in 1 J/cm² increments) |
| Pulse Width | 5ms – 400ms (adaptive) |
| Repetition Rate | Single-shot to 10 Hz (continuous) |
| Cooling System | Quadrasonic: TEC (-10°C to +5°C) + Sapphire contact plate + Deionized water loop (2L reservoir) + Dual radial fans |
| Skin Contact Sensor | Capacitive / Impedance-based with auto-lockout |
| User Interface | 10.4 inch HD Capacitive Touchscreen, real-time fluence mapping & treatment logging |
| Input Power | AC 110-240V, 50/60Hz, 1800VA (max) |
| Dimensions (Console) | 500mm (W) x 600mm (D) x 1150mm (H) |
| Weight | Approx. 45kg (99 lbs) inclusive of handpiece and water coolant |
📥 Download Technical Specification
Click the button below to view or download the full official PDF datasheet.

