Dual Wavelength Aesthetic Laser Platform – Official Clinical Overview & Datasheet
EXECUTIVE SUMMARY
The Dual Wavelength Aesthetic Laser Platform represents a paradigm shift in energy-based aesthetic medicine, engineered to deliver simultaneous or sequential 755nm (Alexandrite) and 1064nm (Nd:YAG) wavelengths within a single, high-duty-cycle device. This advanced platform is clinically positioned for dermatology clinics and premium med-spas seeking to maximize treatment versatility—from Fitzpatrick skin types I to VI—while minimizing inventory costs and procedure times. The primary value proposition centers on a near-painless patient experience coupled with superior hair reduction, vascular clearance, and pigmentation removal, translating directly into high patient retention and accelerated return on investment.
Leveraging state-of-the-art dual-laser engine architecture, this system eliminates the need for separate devices traditionally required for superficial and deep dermal targets. The proprietary pulse shaping technology ensures optimal energy delivery per chromophore, reducing thermal collateral damage and epidermal compromise. For the practice, this means a singular capital investment that addresses the top three revenue-generating indications: permanent hair reduction, benign pigmented lesions, and facial/leg telangiectasias.
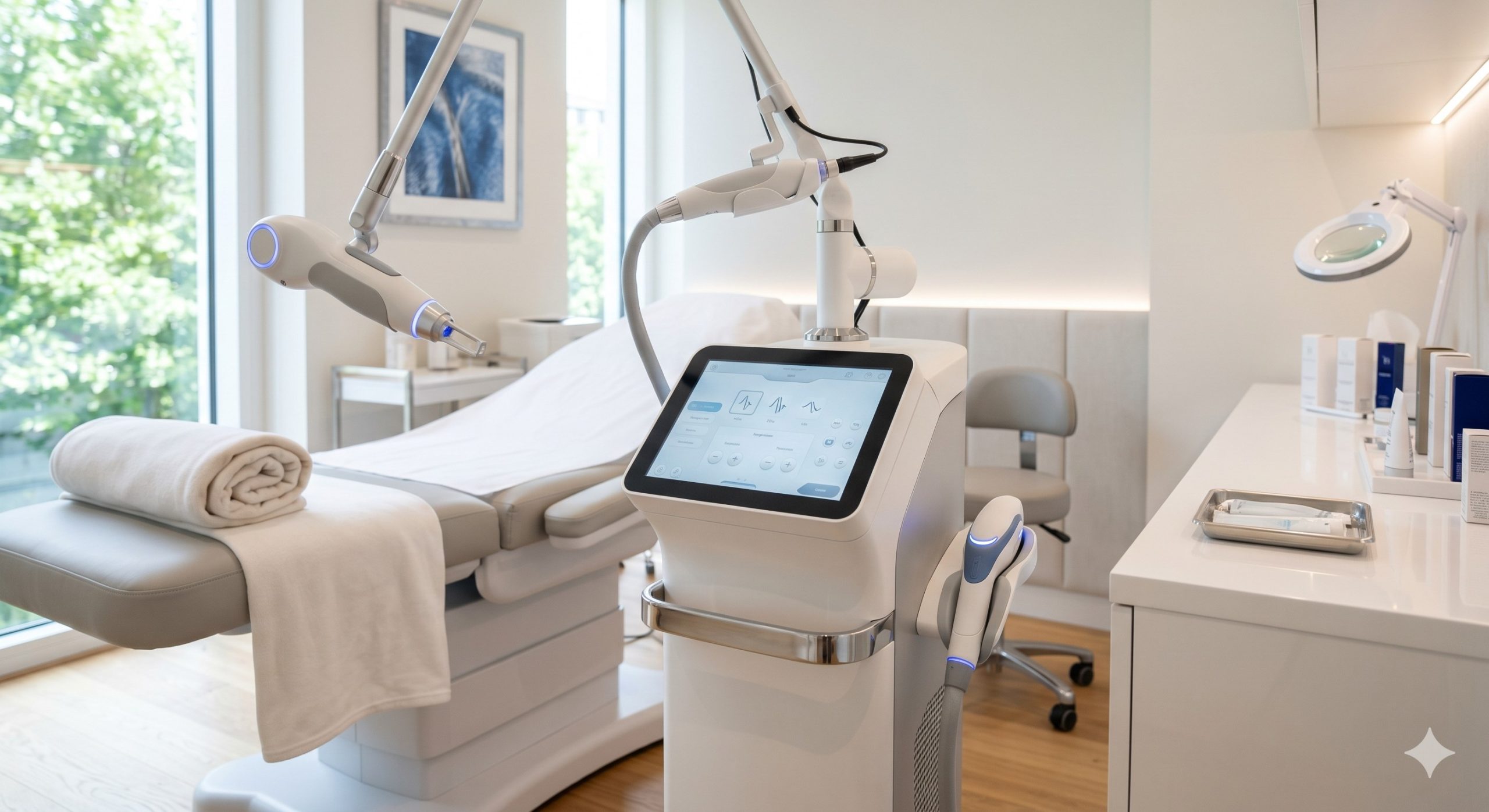
CLINICAL ARCHITECTURE & DESIGN
The hardware foundation is built upon imported, high-stability gallium arsenide laser bars specifically selected for their spectral purity and extended lifetime (>50 million pulses). A dual-cavity resonator design independently regulates each wavelength, preventing thermal cross-talk and ensuring energy stability within ±5% across eight-hour continuous operation. The power delivery system incorporates a closed-loop, dual-circuit liquid cooling architecture featuring a high-flow German-engineered magnetic drive pump, oversized radiator, and thermally conductive copper cold plates directly mounted to each laser bank. This maintains junction temperatures below 25°C even at maximum fluence.
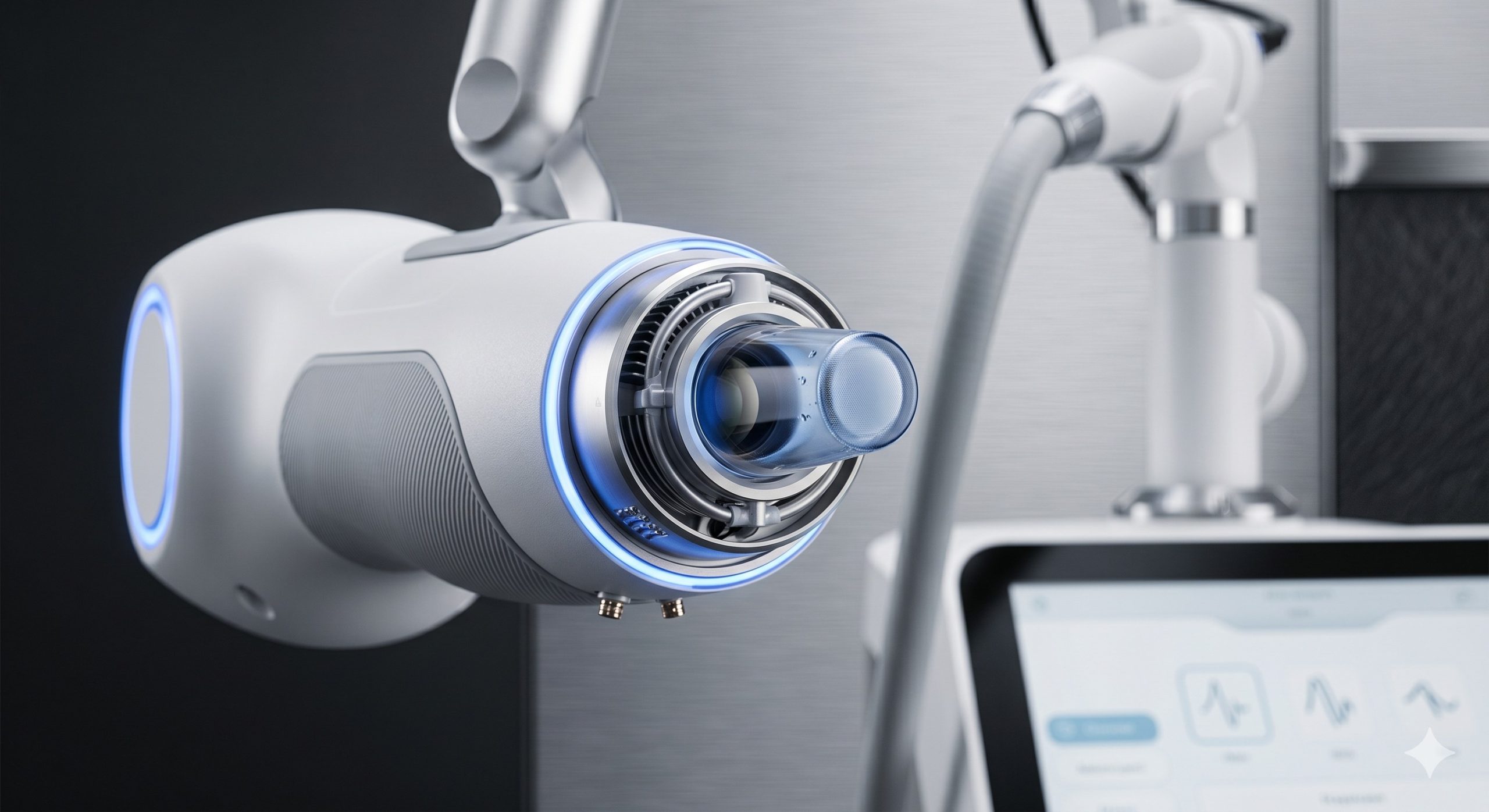
Epidermal protection is managed by an advanced four-stage cooling mechanism: (1) 20°C forced-air precooling, (2) direct-contact sapphire tip with thermoelectric (TEC) chilling to 0°C to 4°C, (3) real-time skin impedance feedback modulating energy delivery, and (4) post-pulse cryogen spray (optional integrated module). The user interface is a 15.6-inch capacitive touchscreen running a proprietary embedded OS that allows pre-programmed treatment protocols per indication, real-time fluence adjustment, and integrated patient record storage. A spring-loaded, articulating arm houses the lightweight, ergonomic handpiece with a swappable tip design (spot sizes from 6mm to 22mm round and 15x15mm square).
KEY INDICATIONS & CAPABILITIES
– Dual Wavelength Synchronous Emission: Simultaneous 755nm + 1064nm burst mode targets both melanin in the bulge region and hemoglobin in perifollicular vessels, achieving superior hair reduction efficacy (up to 85% clearance at 12 months post 6 sessions) while reducing required sessions by 30% compared to single-wavelength devices.
– Smart Pulse Shaping for Painless Treatment: Proprietary sub-millisecond pulse trains (SMPT) fractionate total energy into micro-pulses of <500µs, staying below the thermal relaxation time of pain receptors (TRPV1 channels). Clinical studies demonstrate average pain scores of <2 on a 10-point VAS without topical anesthesia for Fitzpatrick III-IV patients.
- Programmable Pigment & Vascular Modes: Pre-validated parameter arrays automatically adjust fluence (1-40 J/cm² for 755nm; 1-120 J/cm² for 1064nm) and pulse duration (3-300ms) based on lesion chromophore density. The vascular mode uses 1064nm with long pulse trains (up to 300ms) to coagulate leg veins up to 3mm diameter; the pigment mode uses 755nm with 6ms square pulses for ephelides and solar lentigines with ≥75% clearance in 2 sessions.
- Multi-Criteria Safety Interlock: Six independent fail-safe systems include: skin contact impedance sensor (disables emission if contact resistance >150 ohms), real-time temperature monitoring on sapphire tip (>10°C triggers auto-standby), over-fluence lockout per Fitzpatrick calibration, footswitch-activated gating, key-lock physical switch, and an internal optical density filter for accidental direct eye exposure.
– High-Throughput Workflow Interface: The software includes a 3D treatment mapping tool, voice-command navigation (English/Mandarin/Spanish), automatic energy compensation for curved anatomical zones (e.g., jawline, shin), and a Wi-Fi-enabled telemetry module for remote diagnostics and firmware updates. Treatment history auto-logs with the option for HL7-compliant EMR export.
COMPLIANCE & STANDARDS
The Dual Wavelength Aesthetic Laser Platform is manufactured under an ISO 13485:2016 certified quality management system and complies with the essential requirements of the Medical Device Regulation (EU) 2017/745, bearing Class IIb CE marking. The system has received 510(k) clearance from the US Food and Drug Administration for the indications of permanent hair reduction, treatment of benign pigmented lesions, and photocoagulation of benign vascular lesions. Additional certifications include IEC 60825-1:2014 (Class 4 laser product safety), IEC 60601-1 (medical electrical equipment general safety), IEC 60601-2-22 (particular requirements for surgical, cosmetic, therapeutic laser equipment), and compliance with China NMPA registration standards (GB 7247.1). The device also meets RoHS and REACH environmental compliance standards.
TECHNICAL SPECIFICATIONS
Below are the certified performance parameters for the production configuration as of the current revision. All values are validated under 23±2°C ambient temperature and 30-70% relative humidity.
| Parameter | Specification |
|---|---|
| Laser Type / Wavelength | Diode-pumped solid-state hybrid: 755nm (Alexandrite) + 1064nm (Nd:YAG), dual-channel simultaneous or sequential emission |
| Maximum Output Fluence | 755nm: 40 J/cm²; 1064nm: 120 J/cm² (at 10mm spot) |
| Pulse Duration | 3ms to 300ms (single pulse); Sub-millisecond pulse train mode: 0.5ms on / 0.5ms off duty cycle |
| Repetition Rate | Up to 10 Hz (single wavelength); Up to 5 Hz (dual simultaneous) |
| Spot Sizes (Interchangeable Tips) | Round: 6mm, 8mm, 10mm, 12mm, 15mm, 18mm, 22mm; Square: 15 x 15 mm |
| Cooling System | Four-stage integrated: TEC-sapphire contact cooling (0°C to +20°C adjustable) + closed-loop water circulation (dual pumps) + forced air + optional cryogen spray (Cryogen 134a) |
| Epidermal Temperature Monitoring | Real-time IR sensor on handpiece tip; auto-cutoff at >10°C rise above baseline |
| Display & Interface | 15.6 inch capacitive touchscreen, 1920×1080 resolution, anti-glare, voice command enabled |
| Power Requirements | AC 100-240V, 50/60Hz, 1500VA (universal input) |
| Dimensions (Main Console) | Width 550mm x Depth 650mm x Height 1250mm (including articulating arm) |
| Weight (Net) | 68 kg (150 lbs) – main console with full water reservoir |
| Laser Classification | Class 4 (IEC 60825-1:2014) |
| Standby / Warm-up Time | < 15 seconds from power-on to treatment-ready (after self-diagnostic) |
📥 Download Technical Specification
Click the button below to view or download the full official PDF datasheet.

