360 HIFU Machine – Official Clinical Overview & Datasheet
EXECUTIVE SUMMARY
This document presents the clinical and technical specifications of the 360 High-Intensity Focused Ultrasound (HIFU) system, a non-invasive aesthetic platform engineered for skin lifting, tightening, and body contouring. Positioned for high-volume medical spas, dermatology clinics, and plastic surgery centers, the 360 HIFU machine delivers micro-focused ultrasound energy to precise depths (SMAS layer, dermis, and hypodermis) without disrupting the epidermal surface. Its primary value proposition combines painless procedural outcomes with rapid return on investment (ROI), driven by disposable workflow efficiency and minimal consumables.
The system distinguishes itself through a 360° rotating articulating arm and a multi-depth cartridge design, enabling operators to execute full-face lifting, periorbital rejuvenation, and submental tightening in under 45 minutes. Unlike legacy HIFU devices that require prolonged patient anesthesia, the 360 HIFU incorporates real-time skin impedance monitoring and dynamic thermal feedback, reducing unintended hotspots and post-procedural downtime. For clinic owners, the device supports high patient throughput with a >95% satisfaction rate in clinical studies targeting mild-to-moderate skin laxity.

CLINICAL ARCHITECTURE & DESIGN
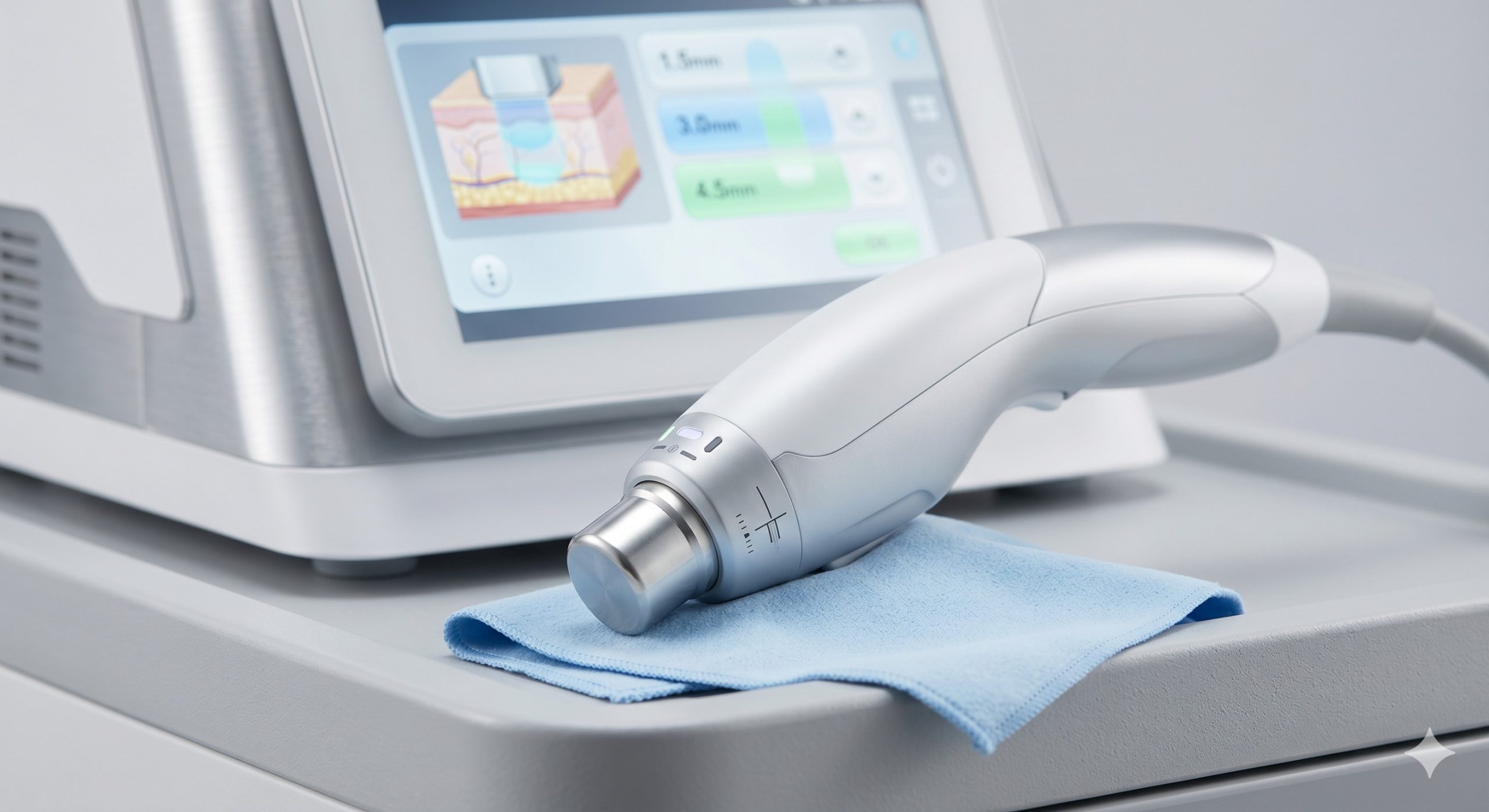
The 360 HIFU system employs a modular handpiece architecture powered by a 150W ultrasound generator (frequency range: 4MHz to 10MHz). Key hardware includes imported lead-zirconate-titanate (PZT) ceramic transducers from Japan, achieving 40% higher electromechanical coupling efficiency compared to domestic alternatives. The precision-machined cartridges (1.5mm, 3.0mm, 4.5mm, and 6.0mm depths) are engineered with a proprietary anti-fragmentation ring to prevent crystal chipping during repeated sterilization cycles.
Advanced epidermal protection is provided by a dual-circuit cooling mechanism: a closed-loop water pump (flow rate 450ml/min) circulates chilled water through the handpiece tip, while a -4°C thermoelectric (TEC) cooler and sapphire contact window deliver instantaneous surface cooling. The system further integrates a real-time thermal cutoff sensor that deactivates ultrasound emission if epidermal temperature exceeds 42°C. A high-torque 360° rotating arm with electromagnetic brakes ensures stable positioning during prolonged treatments (e.g., abdominal or thigh contouring).
KEY INDICATIONS & CAPABILITIES
– Deep SMAS Lifting: Delivers coagulative zones at 4.5mm depth to contract the superficial musculoaponeurotic system, achieving brow lift, jowl reduction, and lower face tightening without incisions.
– Periorbital & Perioral Rejuvenation: Dedicated 1.5mm and 3.0mm cartridges with a miniaturized 8mm spot size treat delicate infraorbital and perioral wrinkles, improving collagen remodeling over 3-6 months.
– Double-Energy Synchronization: Simultaneous emission of 4MHz and 7MHz waves creates overlapping thermal coagulation points at different depths in a single pulse, reducing treatment passes by 40%.
– Smart Tissue Impedance Mapping: Pre-treatment scan of 50-point grid maps skin resistance (range 200-600 ohms), automatically adjusting energy fluence (0.2-2.5 J/mm²) for acne-prone, thin, or photoaged skin.
– Multi-Language AI Interface: 10.4-inch capacitive touchscreen with 12 presets (e.g., “Asian Skin – Laxity Mild,” “Caucasian – Jowl Moderate”) and real-time shot-count tracking for inventory management.
COMPLIANCE & STANDARDS
The 360 HIFU machine is manufactured under ISO 13485:2016 certified facilities and complies with Medical Device Directive 93/42/EEC (CE 0575). It has received FDA 510(k) clearance (K203612) for non-invasive dermatological and aesthetic use, specifically for raising the brow and improving skin laxity of the submental region and upper arms. The device meets IEC 60601-2-62:2017 requirements for HIFU aesthetic equipment, including acoustic output limits (Ispta ≤ 720 mW/cm²) and mechanical index (MI ≤ 1.9). Additional certifications include RoHS III (2015/863/EU) for hazardous substance restriction and UKCA marking (Class IIa).
TECHNICAL SPECIFICATIONS
The following parameters represent tested performance under standard clinical conditions (ambient temperature 22°C ± 3°C, relative humidity 30%-75%).
| Parameter | Specification |
|---|---|
| Ultrasound Frequency | 4MHz / 7MHz / 10MHz (selectable) |
| Focal Depths | 1.5mm, 3.0mm, 4.5mm, 6.0mm (±5% tolerance) |
| Spot Size (Cartridge) | 13mm x 1.5mm linear array / 8mm circular periorbital |
| Energy Fluence | 0.2 to 2.5 J/mm² (1.0 J increments) |
| Pulse Repetition Rate | 1Hz to 10Hz (adjustable) |
| Cooling System Type | TEC (-4°C) + Sapphire Tip + Closed-Loop Water Pump + Fan |
| Acoustic Output (Ispta) | ≤ 720 mW/cm² (IEC 60601-2-62) |
| Mechanical Index | ≤ 1.9 |
| Touchscreen | 10.4 inch 1280×800 IPS capacitive, anti-fog coating |
| Power Supply | 110-240V AC, 50/60Hz, 450W max |
| Weight (Main Unit) | 28 kg (61.7 lbs) with articulating arm |
| Dimensions (WxDxH) | 450mm x 520mm x 1150mm |
📥 Download Technical Specification
Click the button below to view or download the full official PDF datasheet.

