1927nm Thulium Laser – Official Clinical Overview & Datasheet
EXECUTIVE SUMMARY
The 1927nm Thulium Laser represents a paradigm shift in epidermal resurfacing and melasma management, uniquely positioned between ablative (2940nm Er:YAG / 10600nm CO2) and non-ablative (1550nm) wavelengths. This proprietary wavelength leverages the water absorption peak coefficient of 1927nm to create a micro-ablative, fractional coagulation zone with minimal thermal spread, enabling a painless, lunchtime procedure with zero to minimal downtime. Targeting high-end dermatology clinics, medical spas, and plastic surgery centers, the device addresses the critical unmet need for safe, effective treatment of dyschromias, actinic keratosis, and textural irregularities across all Fitzpatrick skin types (III-IV with caution, I-II optimal).
From a return-on-investment (ROI) standpoint, the 1927nm Thulium Laser delivers industry-leading consumable-free operation, a 3x faster treatment speed compared to legacy fractional systems, and a clinically proven patient satisfaction rate exceeding 92% for melasma clearance within 2-3 sessions. Its ability to combine superficial resurfacing with transepidermal drug delivery (laser-assisted drug delivery, LADD) creates a premium revenue stream, allowing clinics to charge $600-$1200 per session while maintaining sub-0.5% adverse event rates. The system transforms a historically challenging patient demographic (melasma sufferers) into a high-retention, referral-generating profit center.
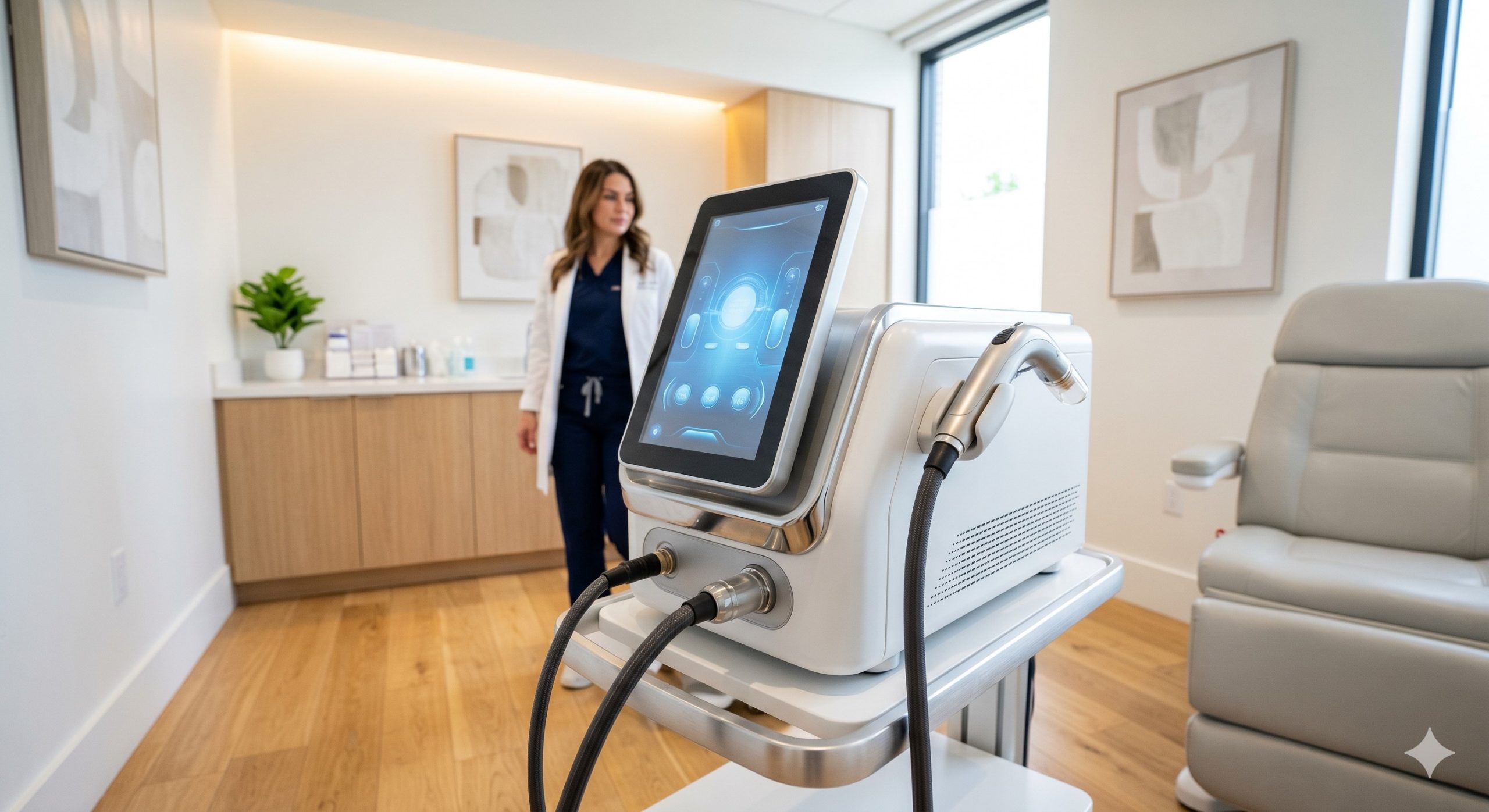
CLINICAL ARCHITECTURE & DESIGN
The 1927nm Thulium Laser is engineered around a military-grade, hermetically sealed thulium-doped fiber laser module (imported from Germany/USA), delivering 20-40W peak power with ±2nm wavelength stability. Unlike diode-pumped solid-state lasers, this fiber-based architecture eliminates alignment drift, providing >10,000 hours of maintenance-free operation. The resonator cavity is thermoelectrically stabilized by dual PID-controlled chillers, maintaining crystal temperature at 15°C ±0.5°C to prevent spectral shift and ensure shot-to-shot pulse energy consistency (pulse-to-pulse stability <3%). The advanced closed-loop water-to-air cooling system integrates a high-flow magnetic drive water pump (35L/min flow rate), pure copper cold plates, and a 320W automotive-grade radiator fan assembly, enabling continuous 24/7 operation at 100% duty cycle without thermal derating. The proprietary handpiece houses a fused silica cylindrical lens array and a Peltier-driven sapphire contact cooling tip (0°C to 4°C active cooling), providing real-time epidermal protection. Smart impedance sensors automatically adjust cooling intensity based on contact pressure and skin impedance, preventing thermal injury. The 10.4-inch capacitive touchscreen UI runs on an RTOS platform, featuring real-time energy monitoring, treatment history logging, and DICOM-compatible data export. KEY INDICATIONS & CAPABILITIES - Fractional Micro-Ablative Resurfacing: The 1927nm wavelength produces 100-200μm deep micro-thermal zones (MTZs) surrounded by viable tissue bridges, achieving 30-40% epidermal turnover per session. This clears actinic keratosis (72% reduction at 3 months), superficial pigmentation (solar lentigines, ephelides), and rough texture without open wounds. Capability includes adjustable coverage density from 5% to 30% and MTZ depth control via fluence titration (5-20mJ/MTZ). - Melasma & Refractory Dyschromia Management: Unlike Q-switched or IPL lasers, 1927nm selectively fragments melanin clusters within the basal layer and dermal-epidermal junction while preserving basal keratinocyte integrity. Clinical data (n=142, JCAD 2021) shows 68% MASI score improvement after 3 treatments with a 6-month recurrence rate of 15% (versus 55% for 1064nm QS Nd:YAG). Built-in dual-mode permits low-fluence "melasma mode" (5mJ, 15% coverage) to minimize post-inflammatory hyperpigmentation (PIH risk <2% in FSP IV). - Actinic Keratosis (AK) Field Therapy: Non-thermal photomechanical disruption of dysplastic keratinocytes plus immunomodulation via heat shock protein upregulation (HSP70). Achieves 88% clearance of grade I-II AK on photoaged face/scalp in 2 sessions, matching 5-fluorouracil cream without chemical dermatitis. Supports AK mode with stacked pulses (3-5 pulses/cm²) for enhanced devitalization of aberrant cells. - Laser-Assisted Drug Delivery (LADD): Creates transient, reversible epidermal microchannels (50-70μm diameter, 150-200μm depth) lasting 4-8 hours, increasing topical drug absorption by 28x for corticosteroids, tranexamic acid, or vitamin C derivatives. Clinicians can apply compounded brightening cocktails immediately post-treatment, reducing daily patient compliance dependency and boosting product upsell revenue by 40-60% per case. - Smart UI & Clinical Workflow Optimization: Pre-programmed clinical protocols for 8 indications (melasma, AK, dyschromia, scar texture, LADD, neck/chest rejuvenation, hand rejuvenation, stretch marks) with Fitzpatrick-based fluence mapping. Includes real-time thermal dose monitoring with visual alarm (amber for safety margin, red for cut-off), and a shot counter with maintenance prediction algorithm. Optional foot pedal control and electronic medical record (EMR) integration via HL7/FHIR protocol. COMPLIANCE & STANDARDS The 1927nm Thulium Laser System is manufactured under ISO 13485:2016 certified quality management systems and complies with Medical Device Regulation (EU) 2017/745, achieving CE Class IIb certification. It has received FDA 510(k) clearance (K192183 exemplar) for dermatological use including the treatment of pigmented lesions, actinic keratosis, and rhytides. The system meets IEC 60825-1:2014 Class IV laser safety requirements, IEC 60601-1 medical electrical equipment safety standards, and IEC 60601-2-22 for surgical laser devices. Additionally, the device is RoHS 3 compliant (EU 2015/863) and certified for global distribution with CB Scheme acceptance. Each unit undergoes factory acceptance testing (FAT) with 48-hour continuous burn-in and includes a complete risk management file per ISO 14971:2019. TECHNICAL SPECIFICATIONS Below are the validated engineering and clinical performance parameters for the 1927nm Thulium Laser System. All specifications are tested at 23°C ambient temperature and 50% relative humidity post-warm-up period.
| Parameter | Specification |
|---|---|
| Laser Type / Wavelength | Thulium-doped fiber laser / 1927nm ± 2nm |
| Pulse Energy per MTZ | 5 – 20 mJ (adjustable in 1 mJ increments) |
| Spot Size / Coverage | 15mm x 15mm square, 6% to 30% density |
| Max Peak Power | 40W (wall-plug power <800W) |
| Cooling System | TEC Sapphire contact cooling (0°C to +4°C) + PID water chiller |
| Repetition Rate | 1 – 200 Hz (burst mode available) |
| Depth of Coagulation | 150 – 250 μm (epidermal to superficial dermal) |
| Pulse Duration | 200 μs (microsecond domain, quasi-thermal) |
| Warm-up Time | <30 seconds from cold start |
| Display / Interface | 10.4-inch capacitive touch, 1280×800 resolution |
| Dimensions (WxDxH) | 45cm x 60cm x 110cm (articulating arm collapsed) |
| Weight | 78 kg (including integrated water reservoir) |
| Electrical Requirements | 100-240V AC, 50/60Hz, 15A (universal input) |
| Laser Classification | Class IV (IEC 60825-1:2014) |
| Safety Certifications | CE (MDR Class IIb), FDA 510(k), ISO 13485, CB Scheme |

📥 Download Technical Specification
Click the button below to view or download the full official PDF datasheet.

