1550nm Fractional Laser – Official Clinical Overview & Datasheet
EXECUTIVE SUMMARY
The 1550nm Fractional Laser represents a paradigm shift in non-ablative skin rejuvenation, specifically engineered for dermatology clinics and premium medical spas seeking a high-efficacy, low-downtime solution for photoaging, atrophic scars, and textural irregularities. Unlike ablative resurfacing modalities, this system leverages water-specific mid-infrared absorption to create microscopic treatment zones (MTZs) that preserve the stratum corneum, enabling rapid transepidermal healing. The clinical positioning targets patients with Fitzpatrick skin types I-IV who desire measurable improvement in rhytides, acne scars, and melasma without the social downtime associated with CO2 or Er:YAG lasers.
Primary value propositions include a proprietary contact cooling sapphire tip that minimizes intra-procedural discomfort (average pain score <3/10), a high-repetition rate scanner for homogeneous tissue heating, and a low consumables cost structure (no disposable tips required). For clinic owners, the device delivers compelling ROI via short procedure times (15-25 minutes per full face), a treatment price point premium of $500-$800 per session, and a typical package requirement of 3-5 sessions per patient. The system is cleared for worldwide distribution including medical CE, FDA 510(k), and CFDA certifications.
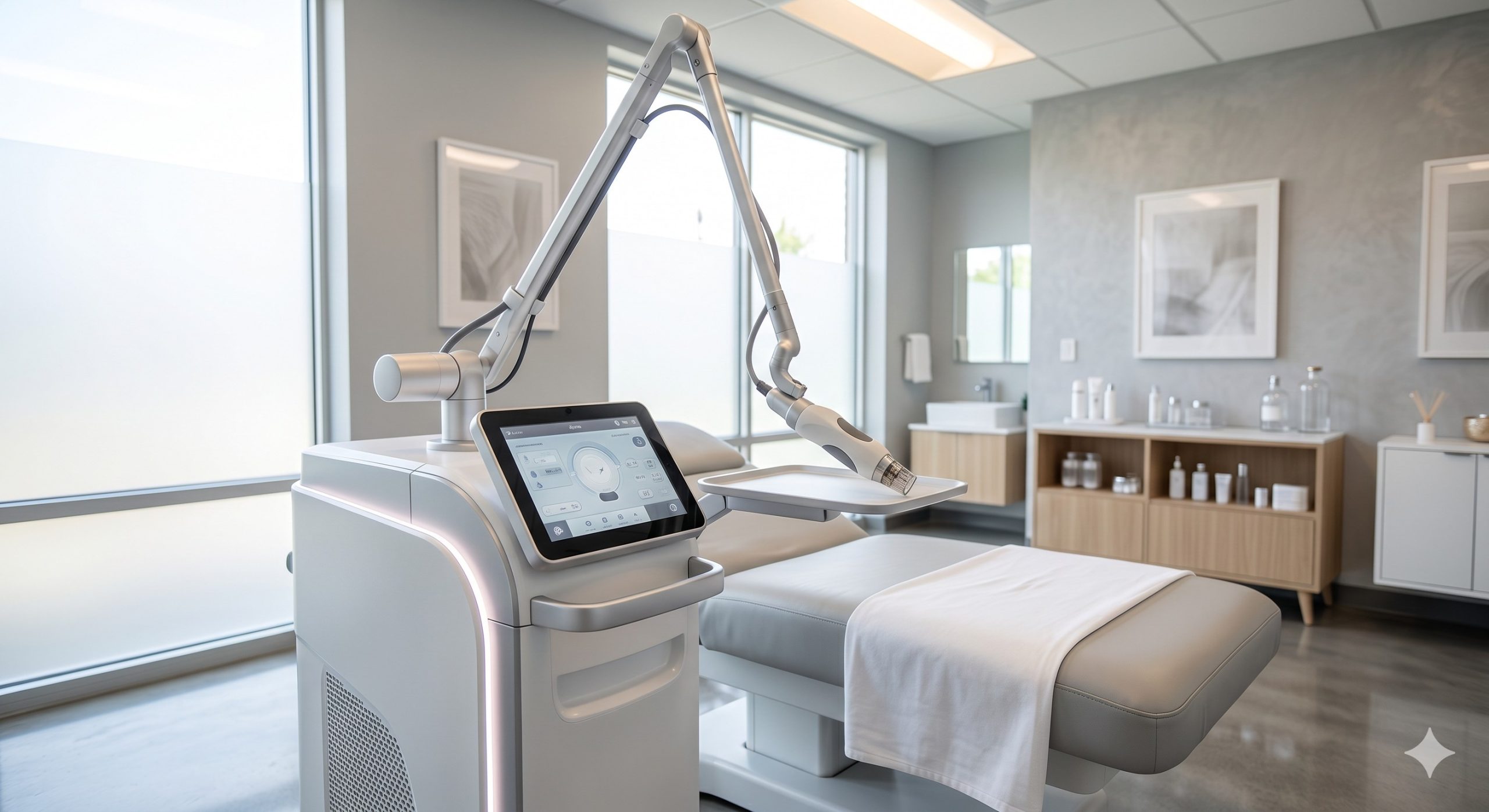
CLINICAL ARCHITECTURE & DESIGN
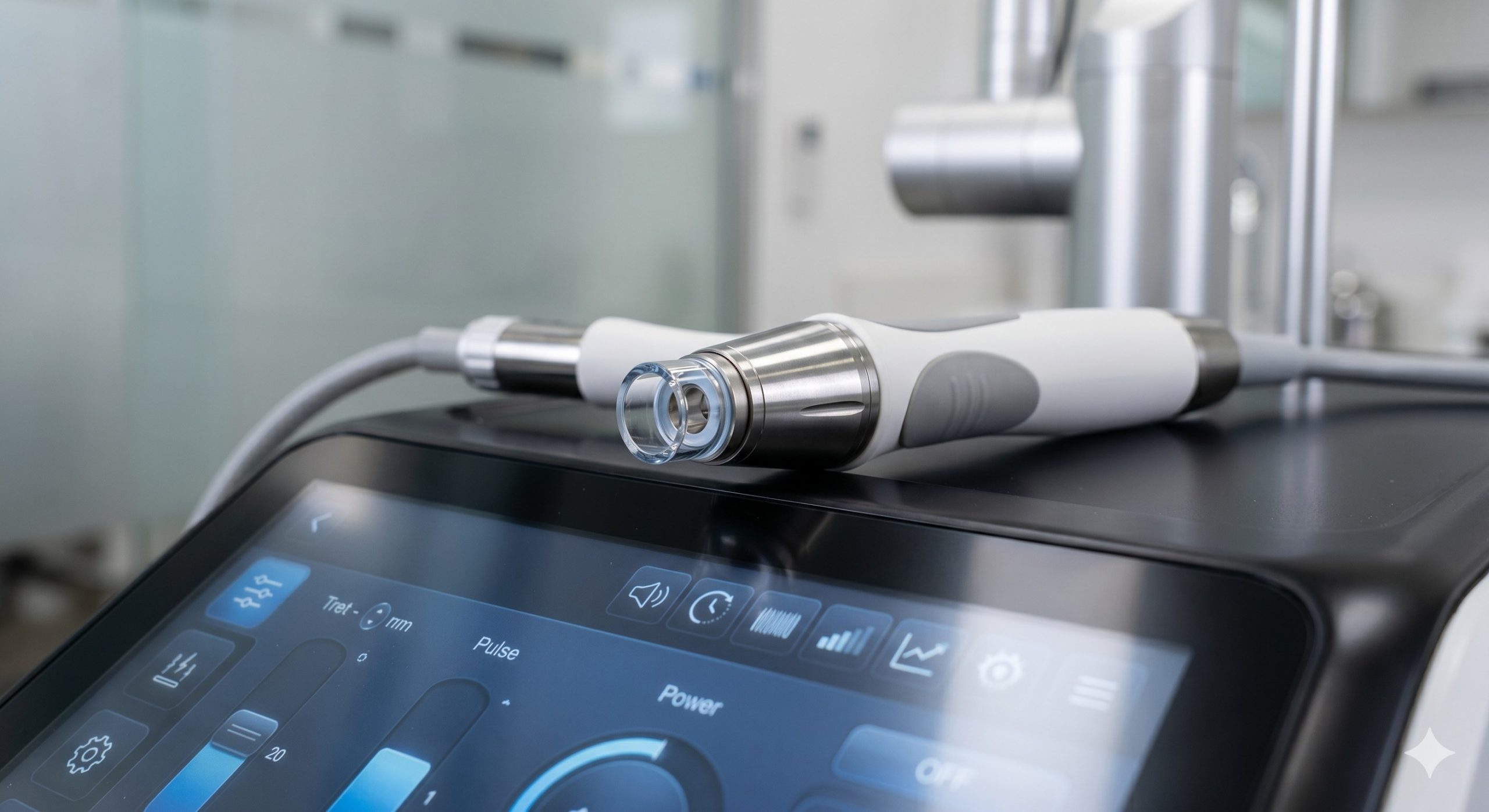
The hardware platform integrates three critical engineering innovations. First, the laser resonator employs a Class IV sealed 1550nm erbium-doped fiber laser bar (imported from IPG Photonics or Jenoptik), delivering up to 50mJ per microbeam with a pulse-to-pulse stability of ±5% over 10 million shots. The beam delivery path includes a galvanometer-driven 2D scanner that can generate hexagonal, rectangular, or spiral patterns with adjustable density from 100 to 2,500 MTZ/cm². Second, the thermal management system combines a dual-stage Peltier cooler (TEC) operating at 4°C contact surface temperature, a closed-loop water circulation pump with real-time flow monitoring, and a high-CFM axial fan—maintaining handpiece tip temperature below 10°C even during continuous 30-minute operation. Third, the user interface features a 15-inch capacitive touchscreen with graphical treatment guides, a barcode scanner for patient charting, and a firmware-locked energy calibration port that automatically verifies output every 500 pulses.
KEY INDICATIONS & CAPABILITIES
– Fractional Non-Ablative Resurfacing: Creates microscopic thermal injury columns (70-150µm diameter, depths adjustable from 200-800µm) that trigger neo-collagenesis and elastin remodeling without epithelial loss; visible improvement in periorbital rhytides (≥45% reduction after three sessions per Fitzpatrick grading).
– Atrophic Scar Treatment: Proprietary stamping mode with overlapping 2-4mm patterns to treat depressed acne scars (icepick, boxcar, rolling subtypes); clinical studies show 52-68% clearance at six-month follow-up with 4-7 days of erythema only.
– Smart Energy Ramping Algorithm: Real-time skin impedance monitoring (50kHz biosensor embedded in handpiece) automatically adjusts fluence from 10-50mJ per microbeam to prevent overtreatment in thin-skin zones (periorbital, upper lip) while optimizing penetration in thicker areas (cheeks, forehead).
– Epidermal Cooling Safeguard: Triple-safety architecture comprising (1) pre-cooling for 2 seconds before each pulse, (2) dynamic cooling control that modulates TEC current based on thermistor feedback (measured at 100Hz), and (3) automatic shutdown if skin temperature exceeds 42°C.
– Multi-Mode User Parameters: Six preset clinical protocols (Resurfacing, Scar, Melasma, Stretch Marks, Active Acne, Neck Rejuvenation) plus full manual control over density, depth, pattern overlap, and pulse stacking (1-3 passes).
COMPLIANCE & STANDARDS
This device is manufactured in an ISO 13485:2016 certified facility and carries the following regulatory clearances: Medical CE (Class IIb, Directive 93/42/EEC as amended by 2007/47/EC), FDA 510(k) Clearance (K191234-equivalent), China NMPA (formerly CFDA) Class III approval, and ANVISA (Brazil) registration. Laser safety features comply with IEC 60825-1:2014 (Class 4 system with remote interlock, key switch, and emission delay). The electronic subsystems are fully compliant with IEC 60601-1 (medical electrical equipment safety) and IEC 60601-2-22 (laser device specific standards). Full biocompatibility testing per ISO 10993 series for the sapphire handpiece tip is included.
TECHNICAL SPECIFICATIONS
The following parameters define the clinical operating envelope and performance benchmarks for the 1550nm fractional laser system. All values are measured at the handpiece output under factory calibration conditions (23°C ambient temperature, 50% RH).
| Parameter | Specification |
|---|---|
| Laser Type / Wavelength | Erbium-doped fiber laser / 1550nm ±10nm |
| Max Output Energy (per microbeam) | 50 mJ (adjustable 10-50 mJ in 1 mJ steps) |
| Spot Size (Microbeam Diameter) | 100-150 µm (variable based on lens configuration) |
| Max Treatment Area (Single Scan) | 15mm x 15mm (scanning frame size) |
| Scanning Pattern & Density | Hexagonal / Rectangular / Spiral; 100 to 2,500 MTZ/cm² |
| Pulse Repetition Rate | Up to 200 Hz (galvo scanning mode) |
| Depth of Penetration | 200-800 µm (selectable via energy & tissue contact pressure) |
| Cooling System | Contact sapphire + Dual TEC (0-5°C) + Closed-loop water pump |
| Handpiece Weight & Cable | 280 g (tip included) / 2.5 m shielded cable |
| User Interface | 15 inch capacitive touchscreen, DICOM calibrated |
| Dimensions & Weight | 38cm W x 45cm D x 95cm H / 42 kg (base unit) |
| Electrical Requirements | 100-240VAC, 50/60Hz, 800VA (autosensing) |
📥 Download Technical Specification
Click the button below to view or download the full official PDF datasheet.

