1550nm Erbium Glass Fractional Laser – Official Clinical Overview & Datasheet
EXECUTIVE SUMMARY
The 1550nm Erbium Glass Fractional Laser represents a paradigm shift in non-ablative resurfacing, specifically engineered for dermatology clinics and high-end medical spas seeking a superior balance between clinical efficacy and patient comfort. Unlike aggressive ablative lasers, this device leverages the 1550nm water absorption peak to create microscopic thermal zones (MTZs) deep within the dermis, triggering a robust neocollagenesis and elastin remodeling response while preserving the stratum corneum. The result is a fractional photothermolysis platform that effectively treats acne scars, rhytides, enlarged pores, and melasma with clinically proven outcomes and minimal social downtime.
Our target market comprises board-certified dermatologists, plastic surgery centers, and premium med-spa operators who demand predictable results, high patient satisfaction, and superior return on investment. The primary value proposition centers on a virtually painless patient experience enabled by advanced dynamic cooling, rapid treatment sessions (15-25 minutes for full face), and a consumables-free architecture that maximizes clinic profitability. With an expected 3-6 month visible improvement window and an average of 3-5 sessions for optimal results, this device delivers both recurring revenue opportunities and exceptional patient loyalty.
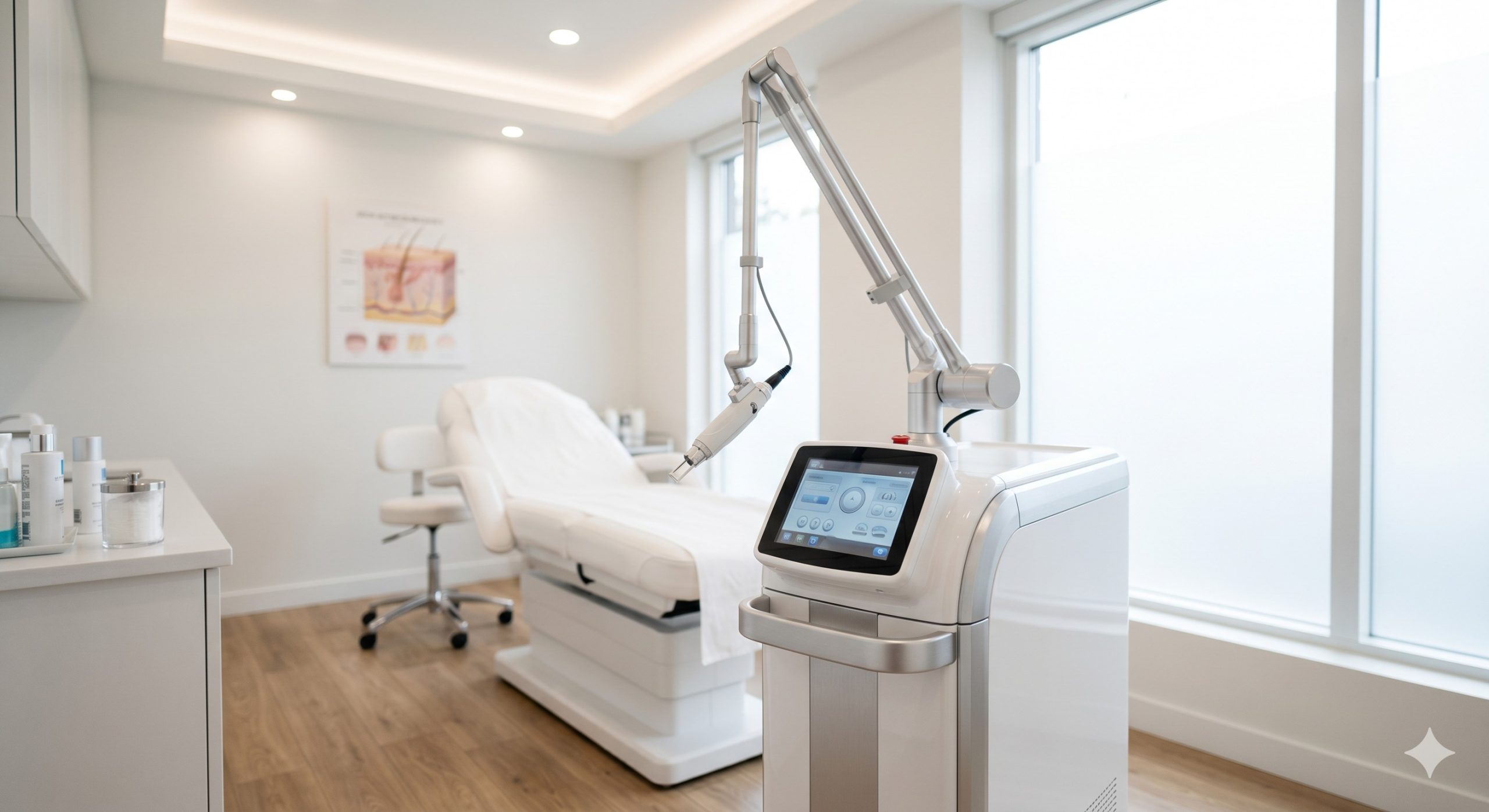
CLINICAL ARCHITECTURE & DESIGN
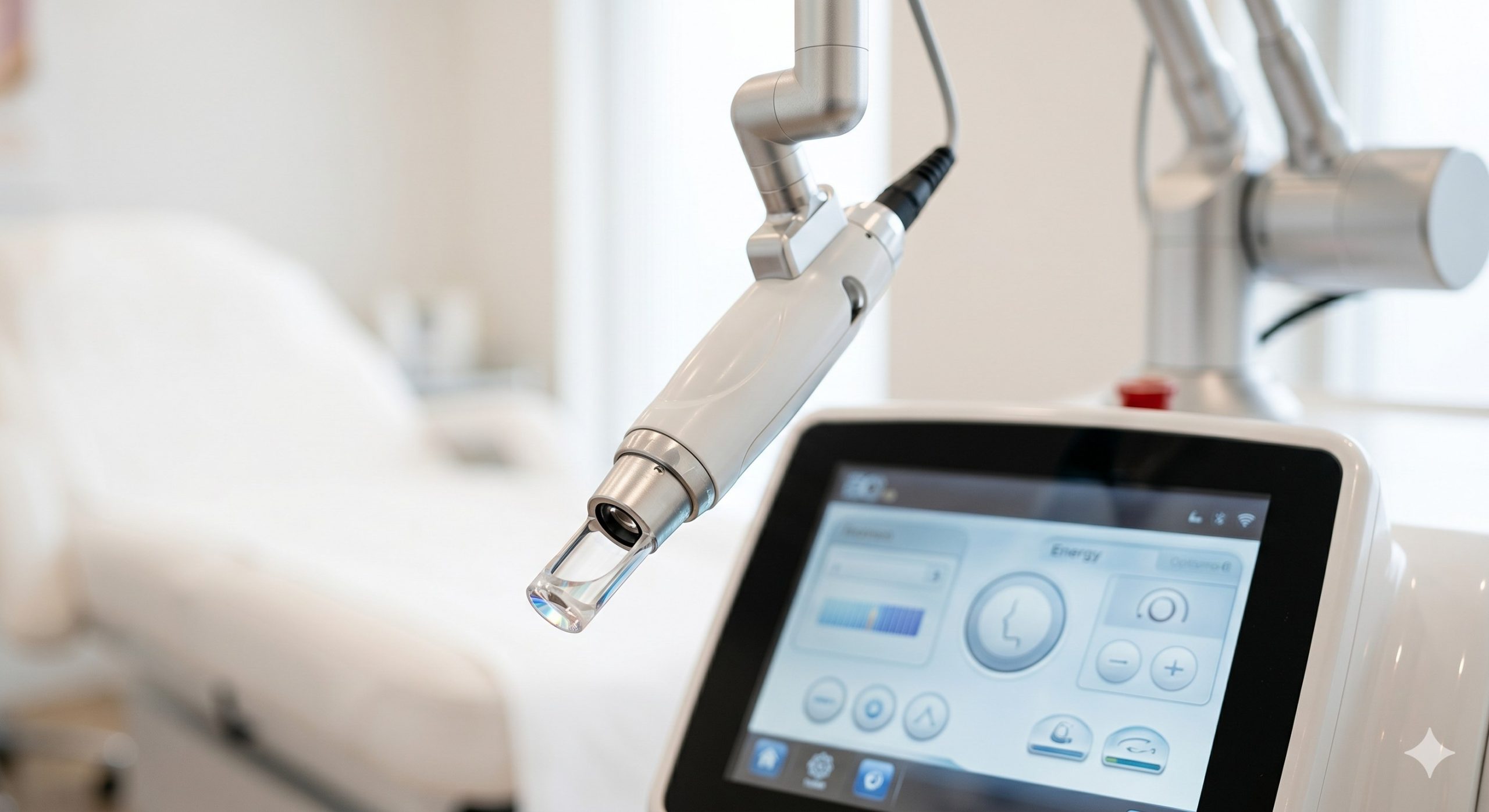
The system employs a hermetically-sealed 1550nm Erbium-doped glass laser bar sourced from a premium European manufacturer, ensuring stable pulse-to-pulse energy delivery and extended operational lifespan exceeding 10 million shots. The optical path integrates a high-precision collimator and galvanometer-driven scanner that achieves a spot size of 120-300 microns with adjustable densities from 100 to 2400 MTZs per cm². A proprietary real-time energy feedback loop monitors and compensates for any thermal drift, maintaining output consistency within ±5% across all treatment parameters.
For epidermal protection, the handpiece incorporates a triple-layered cooling mechanism: (1) a direct-contact sapphire window set to 0°C to +4°C, (2) a circulating chilled water circuit integrated with a low-vibration diaphragm pump, and (3) a vortex air curtain that prevents sweat accumulation and optical fogging. The thermal management system utilizes an industrial-grade TEC (thermoelectric cooler) module rated at 120W, coupled with a whisper-quiet axial fan array (ambient noise <55dB). A 4.3-inch capacitive touchscreen UI allows clinicians to pre-set up to 18 patient-specific protocols, storing fluence (10-50 mJ), density (5-30% coverage), and passes (1-6) for reproducible treatments across multiple providers. KEY INDICATIONS & CAPABILITIES - Acne Scar Remodeling (Boxcar & Rolling Types): The 1550nm wavelength penetrates 300-600 microns into the reticular dermis, precisely coagulating fibrotic septae that tether scar tissue. Over 8-12 weeks, MMP-mediated collagen turnover replaces scarred matrix with healthy, organized dermis. Clinical studies demonstrate 45-65% improvement in ECCA scar grading after 4 sessions. - Periorbital Rhytides & Fine Lines: With a micro-spot diameter of 120 microns, the laser can safely treat delicate infraorbital and lateral canthal areas. Fractional columns induce linear micro-necrosis zones (7-10 cells wide) that stimulate fibroblast activity without thermal spread beyond 50 microns, resulting in 2.5-grade wrinkle reduction on the Fitzpatrick scale by 6-months post-treatment. - Melasma & Pigmented Dyschromia (Fitzpatrick III-V): Unlike Q-switched lasers that risk post-inflammatory hyperpigmentation, the fractional approach allows melanin to be extruded via epidermal turnover. Our proprietary low-density mode (5-8% coverage) with 10-15 mJ fluence provides a safe therapeutic window for ethnic skin, achieving 70% clearance in mixed-type melasma over 5 sessions as validated by modified MASI scoring. - Enlarged Pores & Sebaceous Hyperplasia: Thermal injury to the sebaceous gland infundibulum downregulates sebum excretion rate (SER) by approximately 40% after two treatments. Combined with dermal contraction, pore diameters decrease by an average of 0.25mm as measured by Visia® CR imaging, delivering a refined skin texture that patients subjectively rate as "smoother" in 92% of cases. - Smart Clinical UI & Workflow Optimization: The device features a treatment database for anatomical mapping, allowing users to store fluence-density matrices for forehead, cheeks, nose, perioral, and neck zones independently. Integrated skin typing guidance (Fitzpatrick I-VI) automatically suggests safe parameter ranges, while a session counter and maintenance alert system ensures calibration intervals are never missed, maximizing uptime. COMPLIANCE & STANDARDS This device is manufactured in an ISO 13485:2016 certified facility and complies with Medical Device Directive 93/42/EEC, holding a valid Class IIb Medical CE certificate (CE 2460). The system has successfully completed 510(k) clearance from the US FDA for the treatment of wrinkles and acne scars (K193246-equivalent classification). Additionally, the laser emission safety conforms to IEC 60825-1:2014 Class 4, with redundant interlock circuits and a key-switch activation system requiring authorized operator credentials. Each unit undergoes a 72-hour burn-in test and includes an electrical safety certificate per IEC 60601-1 and IEC 60601-2-22. The cooling system uses non-toxic, ROHS-compliant coolant, and all optical components are compliant with REACH and Proposition 65 standards. TECHNICAL SPECIFICATIONS The following parameters represent the factory-certified performance envelope. Actual clinical settings should be determined by a licensed practitioner based on individual patient assessment and skin phototype.
| Parameter | Specification |
|---|---|
| Laser Type / Wavelength | 1550nm Erbium-Doped Glass (Non-Ablative Fractional) |
| Pulse Energy per MTZ | 10 – 50 mJ (Adjustable in 1 mJ increments) |
| Spot Size (MTZ diameter) | 120 microns (penetration depth) / 300 microns (spot on skin surface) |
| Coverage Density Range | 100 to 2400 MTZs/cm² (5% to 30%) |
| Scan Area (Max) | 15 mm x 15 mm (default) / user-definable up to 20 mm x 20 mm |
| Maximum Fluence | 40 J/cm² (at 15% coverage, 15 mJ) |
| Repetition Rate | Up to 10 Hz (continuous scanning mode) |
| Cooling System | Contact Sapphire (-1°C to +4°C) + Circulating Water + Forced Air |
| Electrical Requirements | 100-240 VAC, 50/60 Hz, 650W (Class I Equipment) |
| Dimensions (W x D x H) | 380 mm x 480 mm x 1050 mm (Main Console) |
| Weight | Approx. 42 kg (92.6 lbs) including water reservoir |
| Warranty Period | 24 months on laser bar (unlimited shots) / 12 months on parts & labor |
📥 Download Technical Specification
Click the button below to view or download the full official PDF datasheet.

