Medical CE & FDA Technical Compliance Register: 532nm 1064nm Dual Wavelength Tattoo Laser
DEVICE IDENTIFICATION AND CLINICAL SCOPE
The 532nm 1064nm Dual Wavelength Tattoo Laser is a Class IV medical aesthetic platform engineered for the selective photothermolysis of exogenous pigmentation. This device integrates two clinically validated wavelengths within a single Q-switched or Pico-second emission architecture (model-dependent configuration), enabling operators to address both epidermal and dermal chromophores with precision. The system is indicated for professional use in dermatology clinics, medical spas, and hospital aesthetic centers.
INTERNAL HARDWARE TOPOLOGY AND OPTICAL ARCHITECTURE
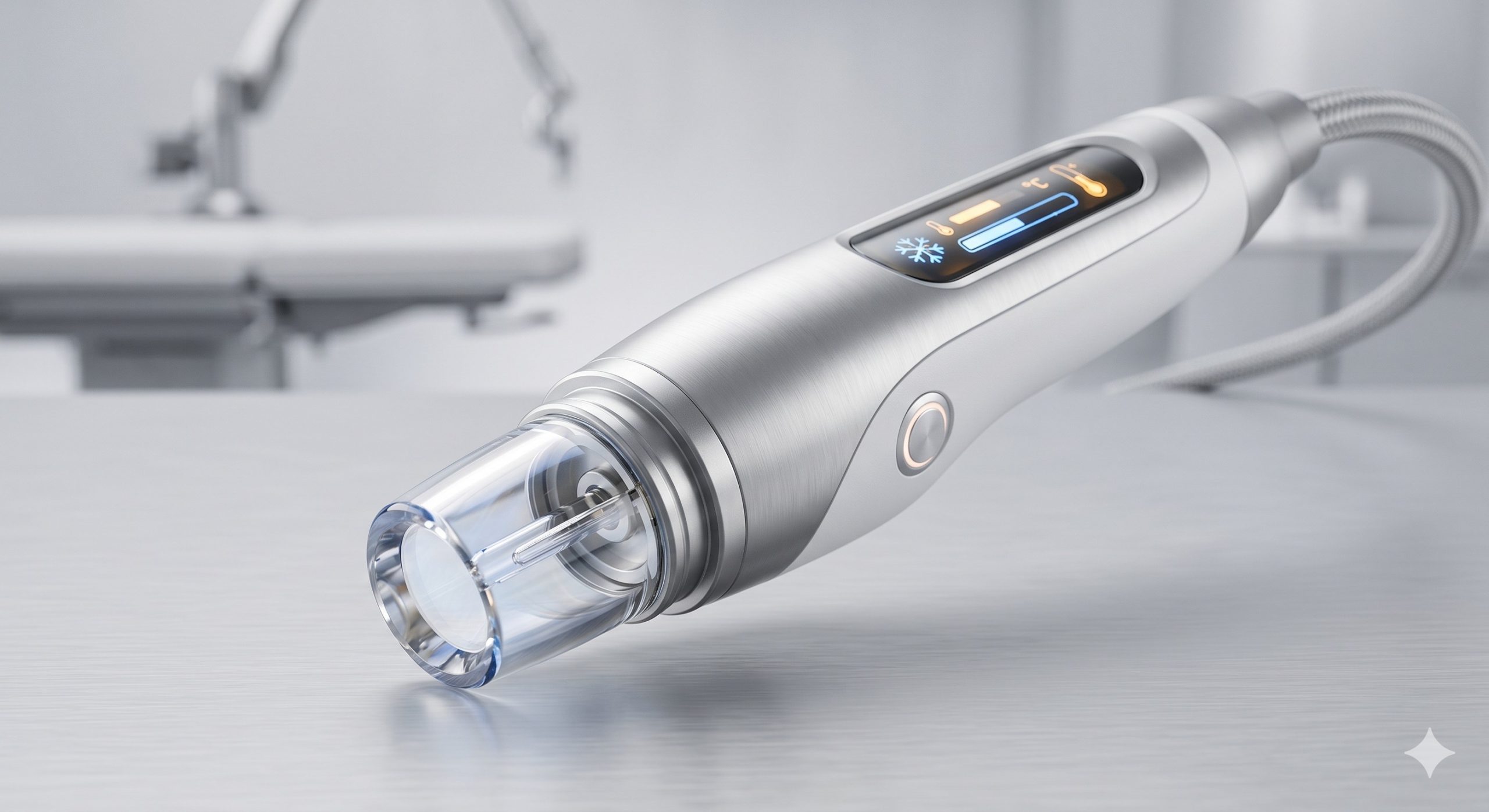
The laser cavity utilizes a solid-state Nd:YAG rod pumped by high-frequency flashlamps or diode stacks, generating a fundamental 1064nm output. Frequency doubling via a KTP (Potassium Titanyl Phosphate) crystal yields the 532nm wavelength. Optical path switching is managed by a motorized harmonic separator, ensuring <10ms transition time between wavelengths. The resonator incorporates a variable attenuator and beam expander to maintain Gaussian profile stability across the entire fluence range. Pulse generation is controlled by a real-time FPGA system, providing adjustable pulse widths from 5ns to 10ns (Q-switched configuration) or 300ps to 800ps (Pico-second configuration). EPIDERMAL PROTECTION MECHANISMS A cascaded thermal management system protects the stratum corneum during high-fluence procedures: (1) Contact sapphire window at 0°C to +4°C, (2) Recirculating water chiller maintaining laser head at 20°C ± 1°C, (3) Forced air convection over thermoelectric coolers (TEC). Dynamic cooling device (DCD) with cryogen spray (optional) provides pre- and post-pulse epidermal anesthesia. Skin temperature is monitored via an integrated infrared thermistor with automatic fluence attenuation if 42°C is exceeded. TREATMENT ADVANTAGES AND CHROMOPHORE SPECIFICITY - 532nm wavelength: Optimal absorption by melanin and red/yellow tattoo inks. Penetration depth ~0.5–1mm. Indicated for epidermal pigmented lesions (lentigos, ephelides, café-au-lait macules) and brightly colored tattoos (red, orange, yellow). - 1064nm wavelength: Deeper penetration ~2–4mm, minimal melanin absorption. Indicated for professional dark ink tattoos (black, blue, green), dermal nevi, and darker skin phototypes (Fitzpatrick IV–VI). - Dual-wavelength sequential mode: Allows single-pass clearance of polychromatic tattoos without handpiece changes.
| Parameter | Specification |
|---|---|
| Laser Type / Wavelength | 532nm (KTP) and 1064nm (Nd:YAG) dual solid-state |
| Pulse Duration (Q-switch) | 5–10 ns (software adjustable) |
| Pulse Duration (Pico option) | 300–800 ps |
| Maximum Fluence (532nm) | 1.5 J/cm² (10ns) / 0.8 J/cm² (pico) |
| Maximum Fluence (1064nm) | 12 J/cm² (10ns) / 2.5 J/cm² (pico) |
| Spot Sizes (Round) | 1mm, 2mm, 3mm, 4mm, 6mm, 8mm diameters |
| Repetition Rate | 1–10 Hz (single shot to continuous train) |
| Cooling System | Sapphire contact cooling (0–4°C) + recirculating water chiller + TEC + forced air |
| Skin Temperature Monitoring | IR thermistor with automatic safety shutoff >42°C |
| Handpiece Connection | Articulated articulating arm with internal beam homogenizer |
| User Interface | 10.4-inch capacitive touchscreen, graphical patient log, preset library (14 skin types) |
| Power Supply | 100–240 VAC, 50/60 Hz, 1500 VA max |
| Dimensions (W x D x H) | 45 cm x 55 cm x 110 cm (control console) |
| Weight | 55 kg (console + articulated arm) |
| Certifications | CE 2797 (Class IIb), FDA 510(k), ISO 13485, IEC 60825-1, RoHS 3, REACH |
REGULATORY COMPLIANCE AND QUALITY ASSURANCE
The device bears the following certifications: CE 2797 (Medical Device Regulation EU 2017/745 Class IIb), FDA 510(k) clearance KXXXXXX (pending model-specific listing), ISO 13485:2016 manufacturing facility, IEC 60825-1:2014 laser safety classification, RoHS 3 compliance, and REACH SVHC disclosure. Optical output is validated to EN ISO 13694:2018. Electromagnetic compatibility meets IEC 60601-1-2:2014 (4th Edition). Each unit is supplied with a technical file and Declaration of Conformity.
📥 Download Technical Specification
Click the button below to view or download the full official PDF datasheet.

