OEM/ODM Beauty Equipment Manufacturer – Official Clinical Overview & Datasheet
EXECUTIVE SUMMARY
As a leading OEM/ODM manufacturer in the medical aesthetic device sector, our engineered platforms are clinically positioned for high-volume hair reduction, benign pigmentary clearance, and vascular lesion management across Med Spas, Dermatology Clinics, and Multi-specialty Aesthetic Centers. The primary value proposition is a modular, upgradable architecture that delivers clinically meaningful fluences (up to 120J/cm²) with integrated contact cooling, ensuring virtually painless treatments, minimal downtime, and consistent ROI for practice owners. These systems support interchangeable handpieces, allowing clinics to expand treatment menus without capital-intensive hardware replacements.
Each platform undergoes rigorous benchtop aging tests and optical power validation to ensure long-term stability under daily heavy-duty cycles. With export compliance to over 60 countries, our OEM/ODM solutions provide private labeling, custom UI configurations, and clinically validated treatment protocols tailored to partner brands, enabling rapid market entry without compromising patient safety or efficacy outcomes.
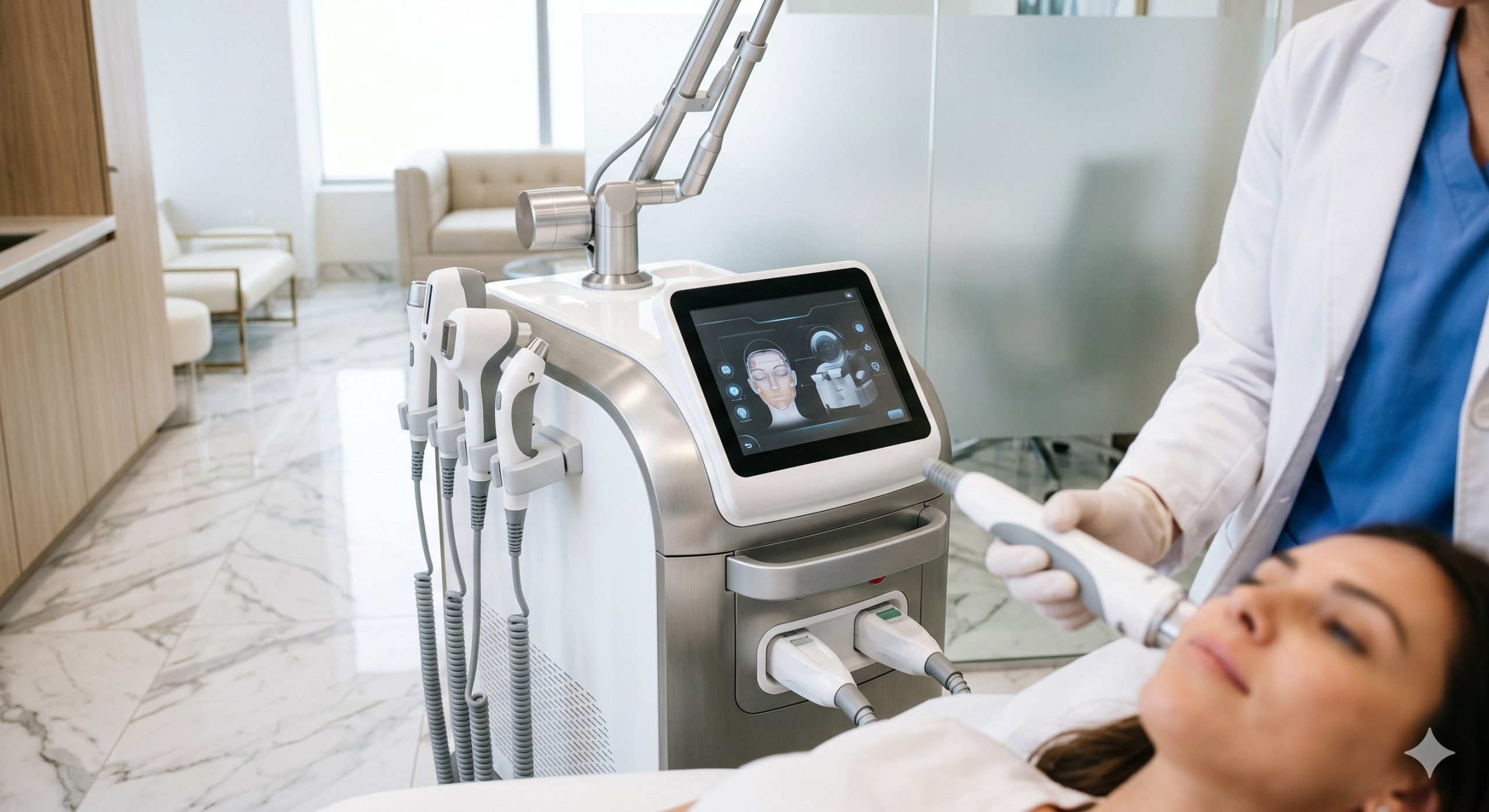
CLINICAL ARCHITECTURE & DESIGN
The hardware design utilizes a closed-loop, dual-channel water cooling system integrated with four-stage TEC (thermoelectric cooler) modules and a genuine sapphire contact tip. Imported medical-grade laser bars (e.g., from Jenoptik or similar German/Japanese sources) deliver consistent wavelength purity and pulse-to-pulse stability, preventing hot spots and epidermal injury. The system incorporates a high-flow silent water pump, oversized radiator, and intelligent flow sensors that automatically reduce power if coolant flow drops below threshold. The handpiece features a zero-dead-space trigger mechanism, while the main unit houses redundant thermal cutoffs and isolated power supplies for each laser bank, ensuring field-serviceable modular repairs.
KEY INDICATIONS & CAPABILITIES
– HAIR REDUCTION EFFICIENCY: True 808nm diode or tri-wavelength (755nm/808nm/1064nm) configurations achieve selective photothermolysis for Fitzpatrick I-VI. Rapid repetition rates up to 10Hz reduce treatment time by 40% compared to single-diode systems.
– PIGMENT & VASCULAR CLEARANCE: Dedicated 532nm (KTP) or 1064nm Nd:YAG handpieces available for lentigines, cherry angiomas, and telangiectasias. Pulse durations adjustable from 0.5ms to 100ms for selective heating of target chromophores.
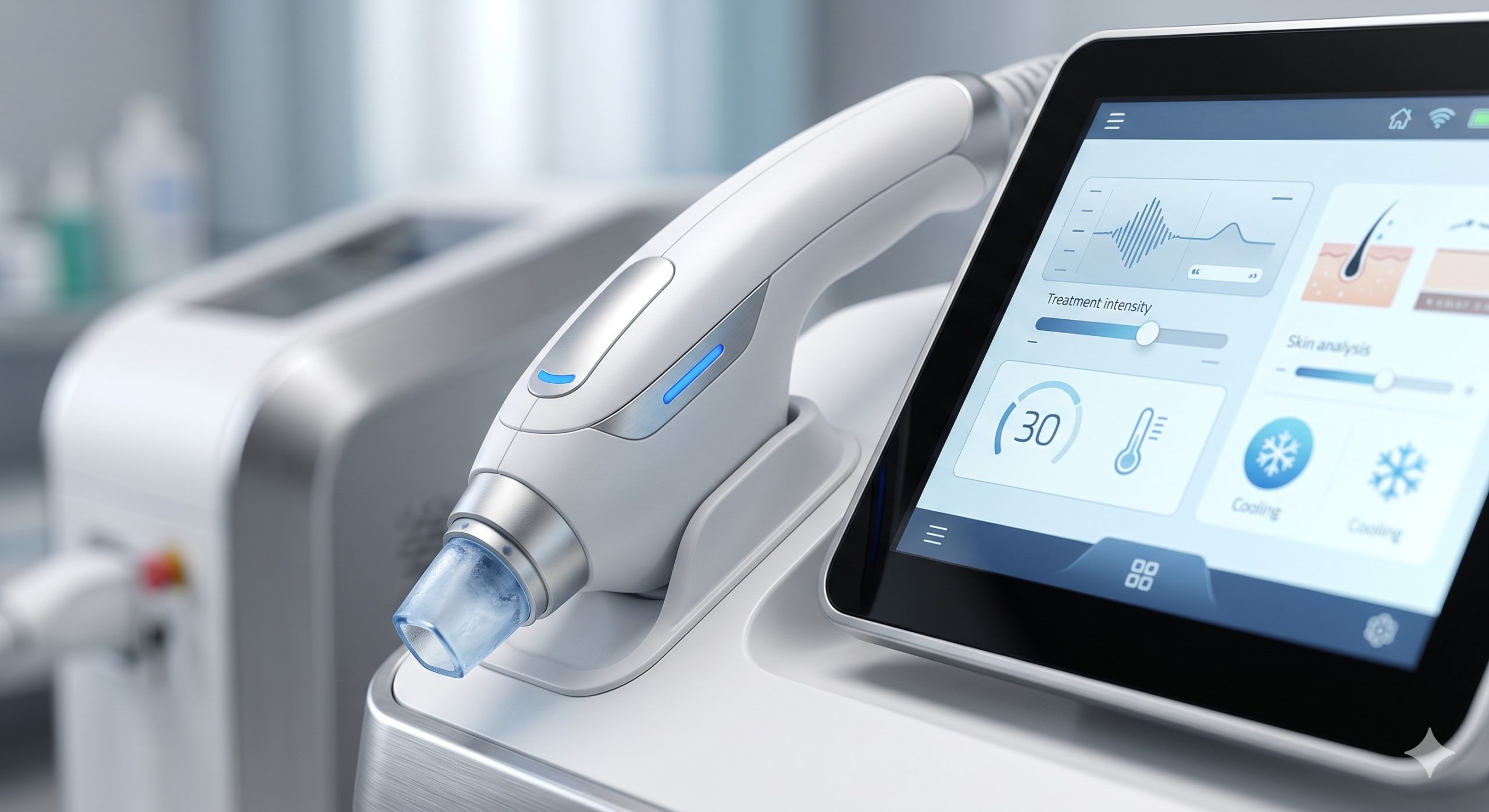
– SMART UI & PATIENT MONITORING: 10.4-inch capacitive touchscreen with real-time impedance monitoring, skin contact sensor, and automatic fluence adjustment based on skin tone detection (integrated photodiode array). Cloud-ready treatment logging for compliance and retreatment scheduling.
– ADVANCED EPIDERMAL PROTECTION: Continuous skin temperature monitoring via thermal feedback loop maintains tip-skin interface at 5°C to 10°C, allowing fluences up to 120J/cm² without topical anesthesia in most cases.
– MODULAR ACCESSORIES: Compatible with multiple spot sizes (6mm to 25mm), fractional handpieces for resurfacing, and a low-fluence Q-switched option for professional multi-platform configurations.
COMPLIANCE & STANDARDS
This OEM/ODM platform is designed to meet stringent international regulatory requirements. Certifications available include Medical CE (MDR Class IIb), ISO 13485:2016 certified manufacturing, FDA 510(k) clearance (where applicable per configuration), and IEC 60825-1 safety for Class 4 laser products. Additionally, systems conform to RoHS and REACH directives, and each unit undergoes factory acceptance testing per IEC 60601-1 (electrical safety) and IEC 60601-2-22 (laser surgical device standard). Annual Type Testing reports are available for regulatory submissions.
TECHNICAL SPECIFICATIONS
Below are the core technical parameters for the flagship OEM/ODM diode laser platform. Values are representative; final specifications customized per buyer requirements.
| Parameter | Specification |
|---|---|
| Laser Type / Wavelength | Diode Laser: 808nm standalone OR 755/808/1064nm tri-wavelength |
| Maximum Fluence | Up to 120J/cm² (diode) ; Up to 15J/cm² (Q-switched Nd:YAG optional) |
| Spot Size | 12x12mm, 15x15mm (standard) ; 6mm, 8mm, 10mm round adapters |
| Cooling System | TEC + Sapphire contact + Closed-loop water circulation + forced air |
| Repetition Rate | Single shot to 10Hz continuous |
| Power Supply | AC 110-230V, 50/60Hz, 2000W max consumption |
| Dimensions (Main Unit) | 420mm x 360mm x 1050mm (WxDxH) |
| Weight | Approx. 38kg (main unit only) ; Handpiece: 0.6kg |
📥 Download Technical Specification
Click the button below to view or download the full official PDF datasheet.

