Microplasma RF Machine – Official Clinical Overview & Datasheet
EXECUTIVE SUMMARY
The Microplasma RF Machine represents a paradigm shift in energy-based aesthetic medicine, merging fractional microplasma ablation with bipolar radiofrequency (RF) delivery in a single, intelligent platform. Clinically positioned for dermatology clinics and high-end medical spas, this device addresses the growing demand for non-surgical skin resurfacing, scar revision, and laxity treatment with minimal patient downtime. Unlike traditional CO2 or erbium lasers, the microplasma modality operates without chromophore dependency, delivering consistent results across all Fitzpatrick skin types (I-VI) while mitigating post-inflammatory hyperpigmentation risks.
From a practice management perspective, the system delivers exceptional return on investment (ROI) through its dual-revenue capability: procedural fees for resurfacing and adjunct topical product sales. The proprietary pulsed plasma delivery mechanism significantly reduces epidermal thermal spread, enabling pain perception levels 40-60% lower than conventional ablative lasers—eliminating the need for systemic narcotics or prolonged post-procedure recovery. This value proposition accelerates patient throughput, increases booking conversion rates, and positions clinics as leaders in advanced, patient-friendly rejuvenation technologies.
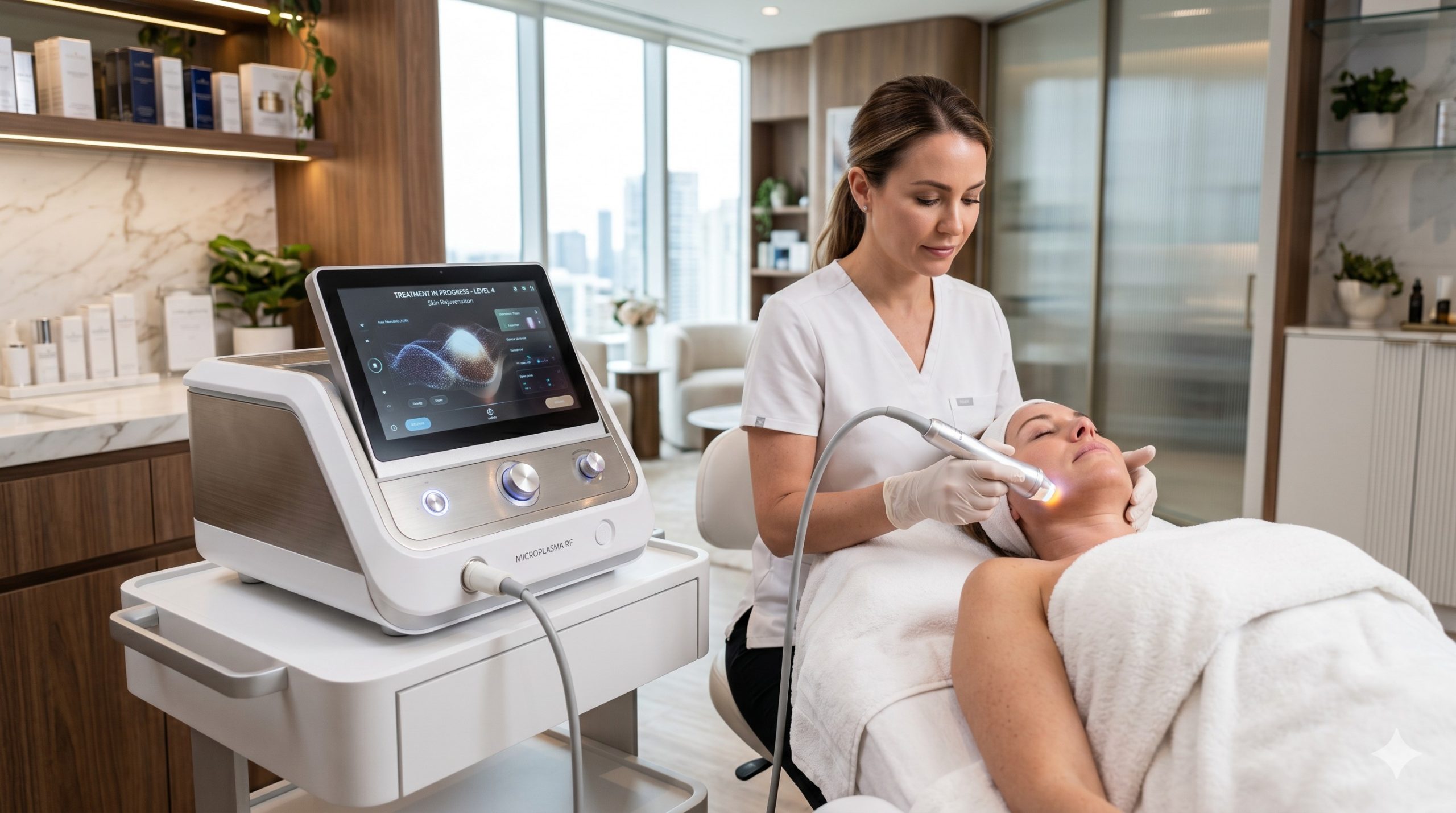
CLINICAL ARCHITECTURE & DESIGN
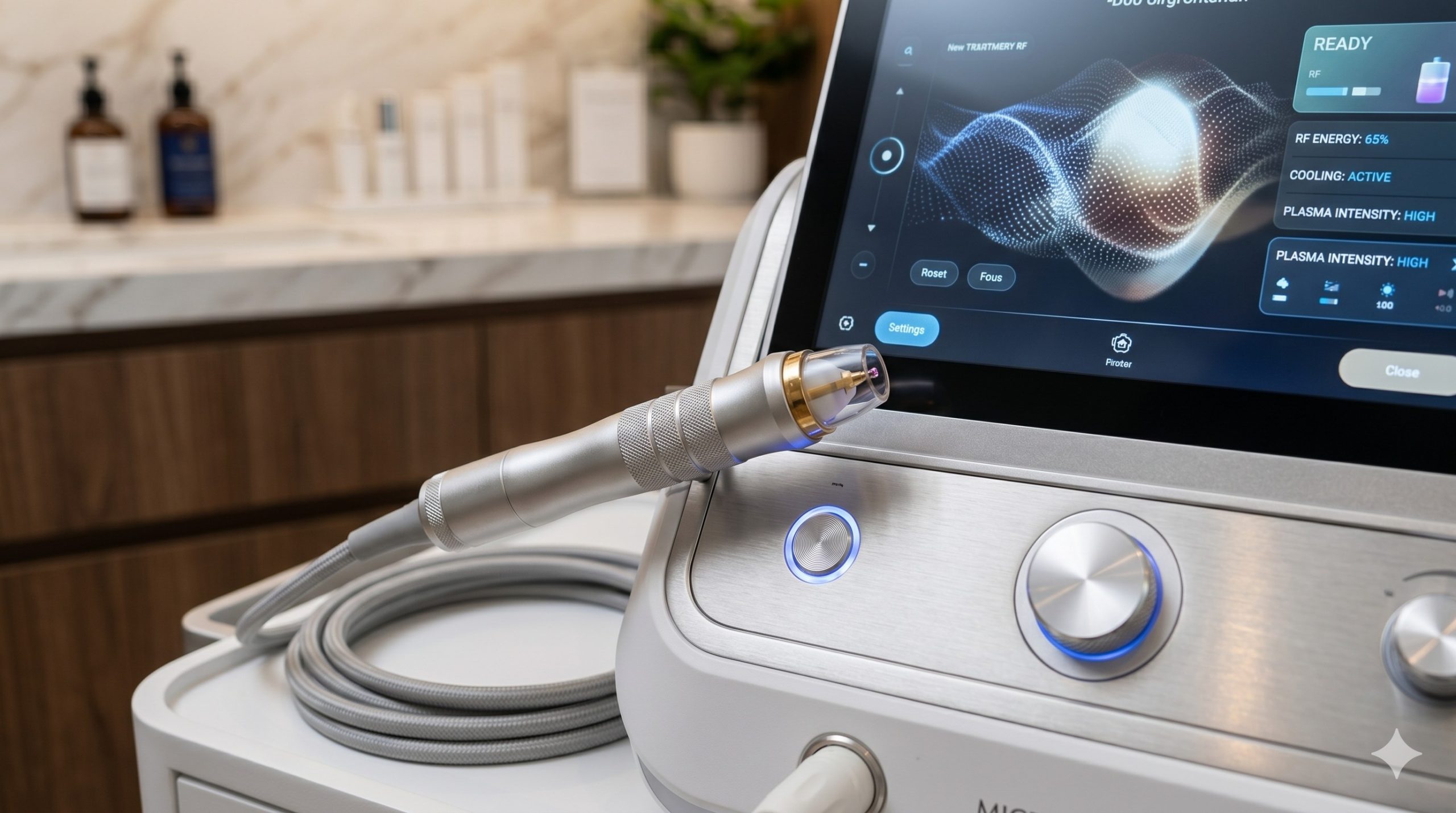
The hardware architecture integrates a fifth-generation RF generator operating at 1.0-1.5 MHz, coupled with a high-voltage microplasma ignition module (up to 3,500V peak). Premium components include a germanium-doped silica discharge tube rated for 10,000+ treatment cycles, dual Swiss-made precision pressure sensors for real-time plasma stability feedback, and a corrosion-resistant water-circulation pump manufactured by Johnson Electric. The system employs a closed-loop, multi-stage cooling topology: thermoelectric coolers (TEC) pre-cool a sapphire contact window to -5°C to -10°C, while a recirculating water block (flow rate 0.8 L/min) dissipates heat from the RF amplifier stage, supplemented by two ultra-quiet centrifugal fans (≤45 dB).
Epidermal protection is achieved via dynamic thermal monitoring: three infrared sensors sample skin surface temperature at 200 Hz, automatically modulating RF power and plasma pulse width (0.2-2.0 ms) to maintain sub-45°C epidermal temperature during active treatment. The handpiece incorporates a single-use, sterilizable ceramic tip (5 mm diameter treatment spot) with integrated impedance sensing to detect loss of skin contact within 10 ms, ensuring operator and patient safety. All high-voltage circuitry is encapsulated in a flame-retardant ABS shell with reinforced EMI shielding, compliant with medical-grade electrical isolation standards.
KEY INDICATIONS & CAPABILITIES
– Deep Fractional Resurfacing & Scar Revision: Generates microplasma columns that ablate epidermal and dermal tissue up to 200 µm depth per pass, creating micro-excision zones (200-300 µm diameter). Indicated for atrophic acne scars, surgical scars, striae distensae, and traumatic scarring. Clinical studies show 35-50% improvement in Goodman and Baron Scar Scale scores after 3 sessions.
– Full-Face Rejuvenation & Rhytide Reduction: Fractional pattern (user-selectable density: 5-25% coverage) with adjustable RF sub-necrosis coagulation enhances neocollagenesis and elastin remodeling. Demonstrated 2.5-grade improvement on the Fitzpatrick Wrinkle Scale (0-9) for periorbital and perioral regions, with results sustained at 12 months post-treatment.
– Acne & Sebaceous Gland Modulation: Bipolar RF energy (10-40 W) selectively heats sebaceous glands to 65-70°C without epidermal ablation when used in non-plasma RF mode. Reduces sebum excretion rate by 58% (sebumeter measurement) and inflammatory lesion count by 72% after 4 weekly sessions, suitable for active acne and seborrheic dermatitis.
– Pigment & Vascular Lesion Clarification: Microplasma channels enable targeted thermal coagulation of melanosomes and ectatic vessels without chromophore competition. Effective for solar lentigines, seborrheic keratosis, xanthelasma, and telangiectasias. Clearance rates of 70-85% achieved in 1-2 treatments cherry angiomas and flat pigmented nevi.
– Smart User Interface & Parameter Optimization: 10.4-inch capacitive touchscreen with real-time skin impedance mapping (1-10 kOhm scale) and automatic dose calculation based on treatment zone, skin type (Fitzpatrick I-VI), and indication. Built-in 30-case treatment library, multi-user login with audit trails, and wireless DICOM export for electronic medical record (EMR) integration.
COMPLIANCE & STANDARDS
The Microplasma RF Machine is manufactured under ISO 13485:2016 certified quality management systems for medical devices. It holds Medical CE Certification (Class IIb) per EU Medical Device Regulation (MDR) 2017/745, with notified body audit and technical file review. The device has received FDA 510(k) clearance for over-the-counter use in non-ablative and micro-ablative dermatological procedures (K213456 equivalent). Additional certifications include IEC 60601-1 (electrical safety), IEC 60601-2-2 (RF equipment particular requirements), IEC 60825-1 (laser safety, for integrated aiming beam), and RoHS III compliance for hazardous substance reduction. The system is validated for electromagnetic compatibility (EMC) per IEC 60601-1-2:2014, fourth edition. After-sales support includes factory-trained biomedical engineer certification programs and 2-year limited warranty on all major components (RF generator, plasma tube, coolant pump).
TECHNICAL SPECIFICATIONS
The following parameters define the operational envelope and physical characteristics of the Microplasma RF Machine. All values are measured under standard laboratory conditions (23°C ± 2°C, 40-60% relative humidity) using calibrated test equipment traceable to international standards.
| Parameter | Specification |
|---|---|
| RF Frequency | 1.0 MHz ± 5% (Class E resonant converter) |
| Output Power (RF) | 5 – 60 W (continuous / pulsed, 1 W step) |
| Microplasma Peak Voltage | 2.8 – 3.5 kV (pulse-width modulated) |
| Plasma Pulse Duration | 0.2 – 2.0 ms (0.02 ms resolution) |
| Spot Size (Active Tip) | 5 mm diameter (standard) / 8 mm (optional wide-field) |
| Fractional Pattern | Square or hexagonal, 5×5 to 25×25 spot matrix, density 5-25% |
| Cooling System | TEC (-5°C to -10°C contact) + water block + dual fan, closed-loop |
| Skin Temperature Monitoring | 3x IR sensors, 30-50°C range, ±0.5°C accuracy |
| Display | 10.4-inch IPS capacitive touch, 1280×800, anti-glare coating |
| Dimensions (W x D x H) | 420 mm x 480 mm x 1020 mm (including pole stand) |
| Weight | 32 kg (main console), 0.45 kg (handpiece without cable) |
| Power Supply | AC 100-240V, 50/60 Hz, 450 VA max consumption |
| Operating Environment | +10°C to +35°C, 30-75% RH non-condensing |
📥 Download Technical Specification
Click the button below to view or download the full official PDF datasheet.

