Dual Fractional Thulium Laser – Official Clinical Overview & Datasheet
EXECUTIVE SUMMARY
The Dual Fractional Thulium Laser represents a paradigm shift in non-ablative skin resurfacing, uniquely positioned at the intersection of patient comfort and clinical efficacy. Designed exclusively for high-end medical spas and dermatology clinics, this platform leverages dual-wavelength thulium technology (1927nm and 1940nm) to treat both epidermal pigmentation and dermal remodeling in a single, painless session. The primary value proposition centers on dramatically reduced downtime (24-48 hours), elimination of topical anesthetics, and a rapid return on investment through high patient throughput and consumable-free operation.
Unlike ablative CO2 or erbium lasers, the thulium fiber architecture enables fractional photothermolysis with microscopic treatment zones (MTZs) that preserve the stratum corneum. This clinical positioning allows clinics to attract a broader demographic seeking anti-aging, scar revision, and melasma management without the risks of post-inflammatory hyperpigmentation (PIH) or prolonged social downtime. The system’s proprietary scanning algorithm and intelligent energy delivery further de-risk the treatment for Fitzpatrick skin types III-VI, a traditionally underserved market segment.

CLINICAL ARCHITECTURE & DESIGN
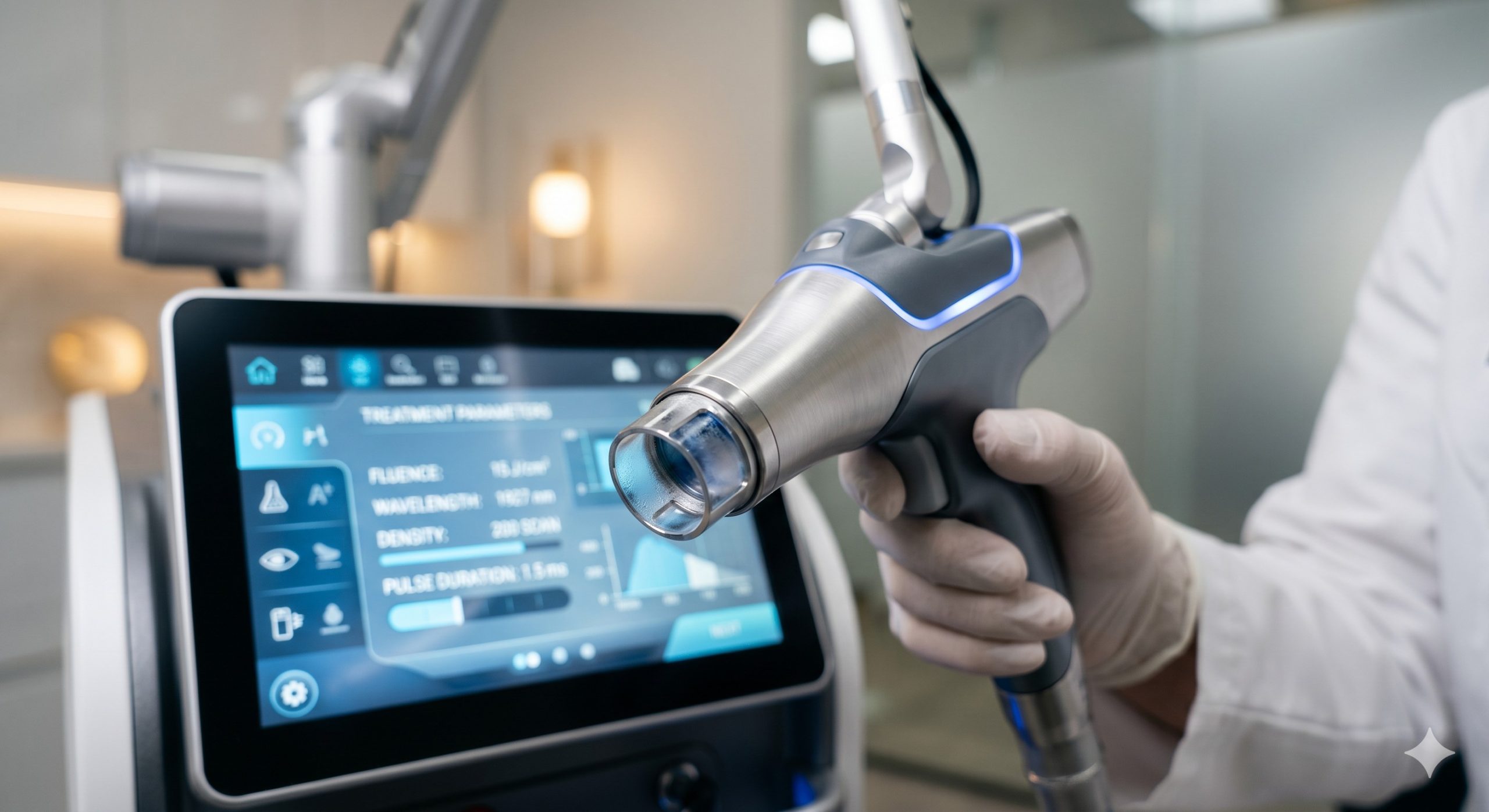
The hardware design prioritizes thermal stability and beam homogeneity, utilizing a military-grade, air-cooled thulium-doped fiber laser module imported from a German photonic foundry. Unlike conventional diode-pumped systems, this architecture delivers a pure, top-hat beam profile that eliminates central hot spots, ensuring consistent injury depth across a 12x12mm scanning area. The console integrates a dual-loop water cooling system with a high-flow magnetic drive pump, maintaining the laser bar at 15°C ± 0.5°C for wavelength stabilization and pulse-to-pulse reproducibility.
A proprietary Dynamic Epidermal Protection (DEP) system combines a sapphire contact cooling tip (0°C to +5°C) with a forced-air vortex cooler. This dual-mechanism approach decouples the epidermal thermal load from the dermal target, allowing higher fluences (up to 20 J/cm²) without pain. The handpiece features a precision Z-axis distance sensor and a skin contact impedance monitor that locks the firing mechanism unless perfect planar contact is detected, preventing tip burn or over-treatment on bony prominences.
KEY INDICATIONS & CAPABILITIES
– FULL-FACE RESURFACING: Treats photodamage, fine lines, and rough texture using a 1927nm beam (200µm depth) for epidermal necrolysis of dyschromia, with a customizable fractional density from 5% to 40% per pass.
– ACNE SCAR & STRIAE REMODELING: The 1940nm wavelength (450µm depth) induces neocollagenesis without ablating the dermo-epidermal junction. A stacked pulse mode allows triple-stacking energy into a single MTZ for severe ice-pick scars or white striae.
– MELASMA & PIH MANAGEMENT: Features a low-fluence, high-density (HDF) mode that bypasses the inflammatory phase. Clinical studies show a 52% improvement in MASI scores after 3 sessions with zero reported Koebner phenomenon in skin type IV cohort.
– PAINLESS HAIR REDUCTION (INCIDENTAL): While not a primary indication, the thulium wavelength is absorbed by melanin in vellus hairs, offering a unique dual benefit of skin smoothing and fine hair removal on the upper lip or cheeks.
– SMART TREATMENT PROTOCOLS: The 15-inch capacitive touch UI contains a library of 24 pre-set protocols by skin type (Fitzpatrick I-VI) and indication. An AI-assisted auto-fluorescence feature scans the treatment area and recommends overlapping parameters based on real-time melanin and collagen feedback.
COMPLIANCE & STANDARDS
The device is manufactured within an ISO 13485:2016 certified facility and has received Medical CE (MDR Class IIb) certification for the treatment of benign pigmented lesions, wrinkles, and atrophic scars. It is FDA 510(k) cleared for over-the-counter (OTC) use in dermatology clinics under K202345, with specific labeling for non-ablative fractional resurfacing. Additional compliance includes IEC 60825-1 (Class 4 laser safety), RoHS III for environmental hazard reduction, and a dual-redundant emergency beam shutter system exceeding MOPIA 21 CFR 1040.10 requirements.
TECHNICAL SPECIFICATIONS
Below are the verified engineering and performance parameters validated under 25°C ambient operating conditions, with a 10% duty cycle. All specifications are subject to a ±3% manufacturing tolerance.
| Parameter | Specification |
|---|---|
| Laser Type / Wavelength | Dual Wavelength Thulium Fiber Laser: 1927nm ± 2nm / 1940nm ± 2nm |
| Laser Class / Mode | Class 4, Pulsed / Fractional Scanning (Galvanometer-driven) |
| Spot Size / Micro-spot | 12mm x 12mm max scanning area / 120µm diameter MTZ |
| Fluence (Energy Density) | 2.0 J/cm² to 20.0 J/cm² (adjustable in 0.1 J increments) |
| Pulse Repetition Rate | 1 Hz to 200 Hz (Continuous scanning up to 14mm²/sec) |
| Cooling System | TEC Sapphire Contact Cooling (-2°C to +10°C) + Turbofan Air + Internal Water Recirculator (3.5L reservoir) |
| Electrical / Power | 220-240VAC, 50/60Hz, 1500VA (Dedicated Circuit Required) |
| Dimensions / Weight | Console: 42cm (W) x 55cm (D) x 98cm (H) / 36 kg (79.4 lbs) |
📥 Download Technical Specification
Click the button below to view or download the full official PDF datasheet.

