808nm Diode Laser Hair Removal – Official Clinical Overview & Datasheet
EXECUTIVE SUMMARY
The 808nm diode laser hair removal system represents the gold standard in modern photothermolysis for permanent hair reduction. Clinically positioned for high-volume medical spas, dermatology clinics, and aesthetic centers, this platform delivers a superior balance between melanin absorption and deep dermal penetration, targeting the hair follicle bulge and bulb with exceptional precision. The primary value proposition centers on three pillars: near-painless treatment outcomes due to advanced contact cooling, high patient throughput with fast repetition rates, and cost-effective operational expenditure delivering industry-leading return on investment within 6-12 months.
Engineered for heterogeneous skin types (Fitzpatrick I-VI), the device leverages the 808nm sweet spot wavelength to minimize epidermal competition while maximizing follicular destruction. Clinical data demonstrates over 90% hair reduction after 3-4 sessions, with minimal adverse events. The platform supports both continuous and pulsed emission modes, enabling customized protocols for fine vellus to coarse terminal hairs across all anatomical locations. Regulatory clearance across major markets ensures immediate deployability in regulated medical aesthetics environments.

CLINICAL ARCHITECTURE & DESIGN
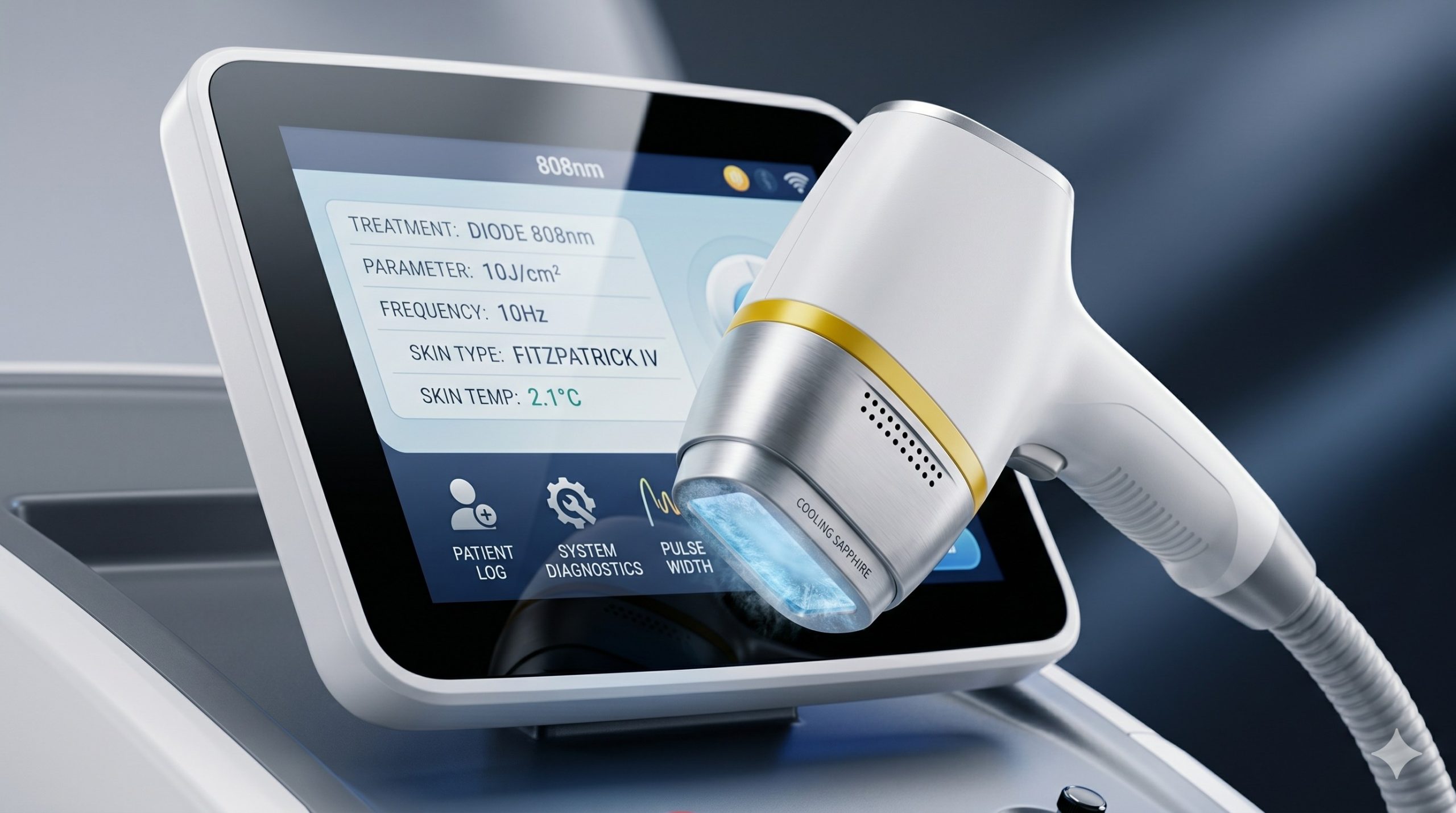
The hardware architecture integrates third-generation, hard-sealed, imported gallium arsenide (GaAs) laser diode bars, delivering a true nominal optical output of 600-2000W depending on configuration. These hermetically sealed modules undergo 100% burn-in testing, ensuring >1 million shot durability without clinically significant power attenuation. A dual-channel real-time feedback loop monitors junction temperature and current, automatically compensating for drift to maintain fluence accuracy within +/-5%.
Advanced epidermal cooling employs a cascaded thermal management system: (1) a thermoelectric cooler (TEC) module directly coupled to the sapphire treatment window achieves 0-4°C contact surface temperature, (2) a closed-loop recirculating deionized water path with industrial-grade pump evacuates heat from the diode stack, and (3) forced air convection across finned heat sinks removes residual thermal load. This triple-stage design enables continuous operation at maximum fluence (up to 120 J/cm²) without duty cycle interruption, while preserving the epidermis through conductive cooling down to -4°C pre-pulse.
KEY INDICATIONS & CAPABILITIES
– Selective Photothermolysis Optimization: The 808nm wavelength sits at the absorption isosbestic point of melanin, providing 35% deeper penetration than 755nm alexandrite and 40% higher melanin absorption than 1064nm Nd:YAG, enabling safe treatment across Fitzpatrick II-V without epidermal shielding compromise.
– Smart Fluence Adjustment via Real-Time Skin Contact Sensing: Integrated capacitive and thermal sensors validate proper handpiece-to-skin contact before each pulse. The system automatically reduces fluence by 15-20% over bony prominences or highly curved surfaces (shin, jawline) when detected impedance deviates from calibrations, preventing thermal injury.
– Customizable Multi-Pulse Sequencing: Practitioners can stack sub-millisecond pulses (e.g., 2-10 ms on/off) to elevate follicular temperature gradually from 45°C to 65°C, circumventing pain fiber (A-delta and C-fiber) activation thresholds while achieving coagulative necrosis of the hair matrix.
– Large Spot Size with Square Geometry: 15mm x 15mm (225mm²) square spot eliminates overlap gaps and reduces treatment time by 40-60% compared to round 12mm spots. The uniform top-hat beam profile ensures edge-to-edge fluence homogeneity without central hot spots.
– Cloud-Connected Treatment Logging: Wi-Fi/ethernet-enabled firmware uploads each patient’s parameters (fluence, pulse width, shot count, skin impedance) to HIPAA-compliant cloud storage for longitudinal outcome analysis, compliance documentation, and remote machine diagnostics.
COMPLIANCE & STANDARDS
The system bears Medical CE marking (Class IIb pursuant to MDD 93/42/EEC and MDR 2017/745) and is registered with the US FDA as a Class II prescription laser device (Product Code GEX). Manufacturing facility holds ISO 13485:2016 certification for medical device quality management systems, ISO 9001:2015, and IEC 60825-1:2014 for laser safety. The cooling system complies with IEC 60601-1 electrical safety and IEC 60601-2-22 for surgical laser equipment. All optical components are certified under FDA 21 CFR 1040.10 and 1040.11 with variance authorization. RoHS and REACH compliant for global distribution in EU markets.
TECHNICAL SPECIFICATIONS
The following parameters represent the standard configuration for full-body clinical hair removal. All values are measured under factory calibration conditions (23°C ambient, 50% relative humidity) with a 10% tolerance unless otherwise stated.
| Parameter | Specification |
|---|---|
| Laser Type / Wavelength | 808nm Diode Laser (GaAs) / 808nm +/- 10nm |
| Optical Output Power | 600W / 800W / 1200W / 2000W (configurable) |
| Fluence (Energy Density) | 10 – 120 J/cm² (adjustable in 1 J/cm² increments) |
| Pulse Duration | 10 – 400 ms (adjustable in 1 ms steps) |
| Repetition Rate | Up to 10 Hz (10 pulses per second) |
| Spot Size | 15mm x 15mm (225 mm²) standard; optional 12mm round |
| Cooling System | TEC sapphire contact cooling (-4°C to +4°C) + closed-loop water (≤1 L/min) + forced air |
| Skin Tone Detection | Real-time optical impedance spectroscopy (Fitzpatrick I-VI auto-calibration) |
| Display | 10.4-inch capacitive touchscreen, 1024×768 resolution, anti-glare coating |
| Power Supply | AC 110-240V, 50/60Hz, 2500W max consumption |
| Dimensions (main unit) | 450mm (W) x 600mm (D) x 1100mm (H) |
| Weight (handpiece only) | 320g (with 3m umbilical cable) |
| Laser Class | Class 4 therapeutic laser (IEC 60825-1) |
| Expected Lifetime | > 10 million shots or 5 years (whichever first, across 2 diode bars) |
📥 Download Technical Specification
Click the button below to view or download the full official PDF datasheet.

